Solvent‑Free, Folate‑Functionalized Nanoneedles for Targeted Delivery of 10‑Hydroxycamptothecine
Abstract
Traditional nanoparticle formulations of hydrophobic anticancer drugs often rely on organic solvents, raising concerns about residual toxicity. Here, we present a fully green synthesis of folate‑modified 10‑hydroxycamptothecine (HCPT) nanoneedles (HFNDs) that eliminates organic solvents while achieving high drug loading (70.2 %) and a robust targeting capability. In vitro, HFNDs demonstrate superior cellular uptake in HeLa cells compared to non‑targeted chitosan‑HCPT nanoneedles, and MTT assays reveal enhanced cytotoxicity at equivalent HCPT doses. In vivo imaging confirms preferential tumor accumulation of HFNDs, and therapeutic studies in HeLa xenografts show pronounced tumor suppression with minimal systemic toxicity. This solvent‑free approach offers a promising platform for safe, efficient chemotherapeutic delivery and imaging.
Background
Over the last two decades, research has focused on overcoming the pharmacokinetic limitations of conventional chemotherapy through nanoparticle‑based delivery systems that provide prolonged circulation, active targeting, and multimodal imaging capabilities [1, 2]. Recent advances have highlighted the importance of particle shape, with high‑aspect‑ratio nanostructures showing enhanced cellular internalization and tumor penetration [10, 26]. However, most of these strategies still depend on organic solvents to solubilize hydrophobic drugs, leading to residual solvent concerns that can accumulate in the body and pose safety risks [27]. Our study addresses this gap by developing HCPT‑loaded, folate‑modified nanoneedles via a completely green, solvent‑free method, thereby combining high drug loading, targeted delivery, and imaging potential.
Methods
Materials
All reagents were analytical grade and used as received. Deionized water was employed throughout. FA was sourced from Bio Basic Inc.; HCPT (> 99 % purity) from Lishizhen Pharmaceutical Co.; chitosan (Mw = 70 kDa, 90 % deacetylation) from Zhejiang Aoxing; NHS and EDC from Sigma‑Aldrich.
Synthesis of the FA‑Chitosan Conjugate
FA (10 mg), chitosan (20 mg), EDC (4 mg), and NHS (4 mg) were stirred in 2 mL PBS (pH 5.5) at room temperature for 12 h. The mixture was dialyzed against pH 10 buffer to remove excess FA, then centrifuged (5 000 rpm) and lyophilized for 24 h to yield CS‑FA powder.
Preparation of HFNDs
HCPT (10 µg) was dissolved in 200 µL 0.1 M NaOH (solution A); CS‑FA (10 µg) in 200 µL 0.1 M HCl (solution B). Solutions A and B were added dropwise to 1 mL pure water under vigorous stirring for 30 s, then sonicated (200 W, ice bath) for 6 min. The resulting suspension was centrifuged (10 000 rpm, 5 min) and lyophilized for 24 h. For comparison, chitosan alone replaced CS‑FA in solution B to produce non‑targeted nanoneedles (NDs).
Characterization
SEM (UV‑70, 15 kV) visualized morphology. Particle size and zeta potential were measured with a Malvern Zetasizer Nano‑ZS. XRD (X’pert PRO) assessed crystallinity. FA content was quantified by UV at 281 nm; HCPT loading by fluorescence at 383 nm. Loading and entrapment efficiencies were calculated using equations (1)–(4) below.
$$\begin{aligned} \text{Drug loading %} &= \frac{\text{weight of HCPT in HFNDs}}{\text{weight of HFNDs}}\times100\%\;(1)\\ \text{Entrapment efficiency %} &= \frac{\text{weight of drug in HFNDs}}{\text{weight of feeding drug}}\times100\%\;(2)\\ \text{FA % in conjugation} &= \frac{\text{weight of FA in conjugation}}{\text{weight of conjugation}}\times100\%\;(3)\\ \text{FA loading %} &= (1-\text{HCPT loading %})\times\text{FA % in conjugation}\times100\%\;(4) \end{aligned}$$In Vitro Drug Release Study
HFNDs were dispersed in 10 mL PBS and placed in a 3 kDa MWCO dialysis bag, then immersed in 200 mL PBS (pH 7.4) at 37 °C, 100 rpm. Samples were collected over 48 h and analyzed by fluorescence spectroscopy.
Confocal Imaging of Cells
HeLa cells were seeded and incubated 24 h before exposure to HFNDs for 8 h. Imaging was performed on a Leica confocal microscope (HCPT excitation 382 nm, emission 500–550 nm).
Cellular Uptake Measured by Fluorescence
After 6 h incubation with NDs or HFNDs (equivalent HCPT), cells were washed, digested with trypsin/EDTA, and sonicated. HCPT content was quantified by fluorescence at 382 nm.
Cytotoxicity Assays
MTT assays were conducted on HeLa cells seeded in 96‑well plates. After 24 h drug exposure, 20 µL of 5 mg/mL MTT was added for 4 h, followed by 150 µL DMSO. Absorbance was read at 570 nm.
Biodistribution
DiR‑loaded NDs and HFNDs were injected IV into HeLa‑tumor‑bearing nude mice. Fluorescence imaging was performed with a Maestro system at 1, 4, 8, 12, and 24 h. Ex vivo imaging of tumor and major organs was conducted at 24 h.
Tumor Inhibition In Vivo
When tumor volumes reached ~60 mm³, mice received IV injections of saline, free HCPT, NDs, or HFNDs (80 µg HCPT per mouse) every 3 days. Tumor volume (0.5×L×W²) and body weight were monitored for 21 days. Post‑mortem, tumors were weighed and H&E‑stained for histology.
Statistical Analysis
Data were analyzed by two‑tailed Student’s t‑test; *p* < 0.05 indicated significance (95 % confidence).
Results and Discussion
Synthesis of the FA‑Chitosan Conjugate
FA was covalently linked to chitosan via an EDC/NHS‑mediated amidation (Fig. 1). FT‑IR spectra (Fig. 2) revealed a pronounced C=O stretch at 1605 cm⁻¹, confirming amide bond formation. UV quantification yielded an FA loading of 23.4 ± 2.5 % in the conjugate.
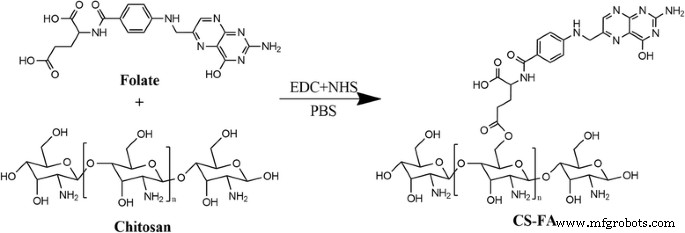
Synthetic route of CS‑FA conjugate
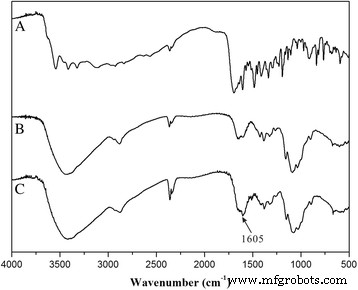
FTIR spectra of (a) FA, (b) chitosan, and (c) CS‑FA
Preparation of HFNDs
HCPT’s aqueous insolubility and chitosan’s acid solubility were exploited: HCPT dissolved in NaOH and CS‑FA in HCl. Neutralization of the two solutions produced a poor solvent for both species, triggering HCPT precipitation and concurrent CS‑FA encapsulation into nanoneedles (Fig. 3a). Ultrasound and CS‑FA’s steric stabilization prevented bulk crystal growth, yielding high‑aspect‑ratio nanoneedles (Fig. 3b–c). A design‑of‑experiments screen (Table 1) optimized the HCPT:CS‑FA ratio, ultrasonic power, pH, and concentration to achieve the desired morphology.
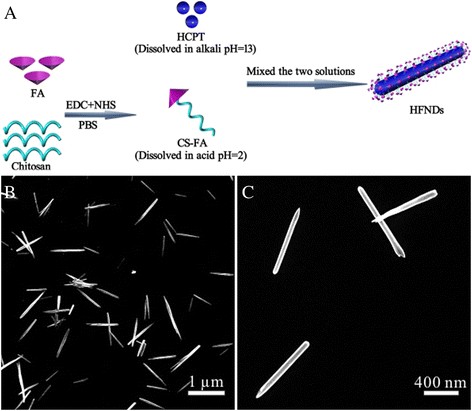
a Illustration of the green synthesis; b, c SEM images of HFNDs
SEM analysis (Fig. 4) showed that excess CS‑FA coated the particle surface, while insufficient CS‑FA led to aggregation. Neutral pH was essential to preserve nanoneedle integrity, and ultrasonic power influenced aggregation and fragmentation. Higher mixture concentration reduced particle size, as confirmed by DLS (Fig. 4f). The optimized HFNDs displayed an average length of ~800 nm, width ~80 nm, hydrodynamic diameter 104.3 ± 5.7 nm, and a +16.3 ± 1.9 mV zeta potential. The 2 wt % dispersion remained stable for ≥2.5 days.
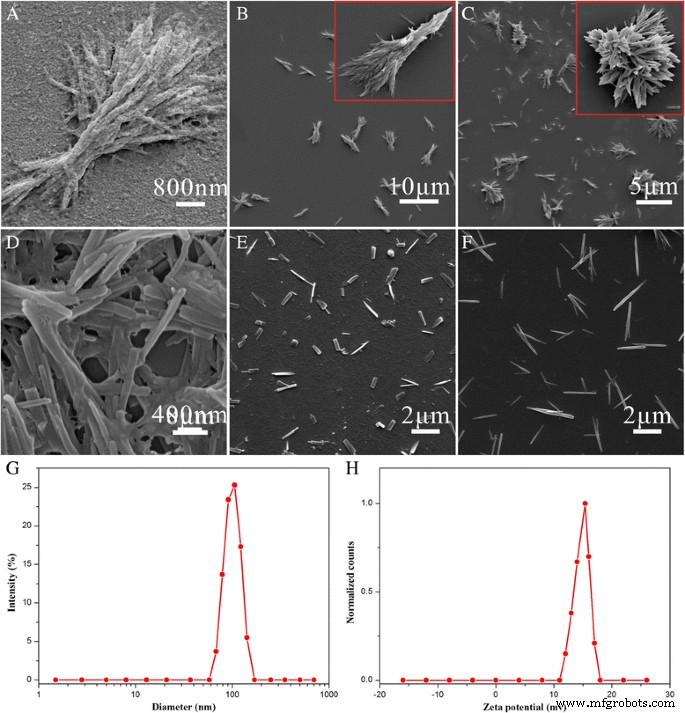
a–f SEM images under varied conditions; g size distribution; h zeta potential
Drug loading was 70.2 ± 3.1 % and encapsulation efficiency 83.1 %. FA accounted for 7.0 % of the total mass, calculated from the conjugate’s FA percentage.
XRD Analysis
XRD patterns (Fig. 5) confirmed that HCPT remained crystalline within HFNDs, while the semi‑crystalline chitosan contributed broad peaks. In contrast, bulk HCPT crystals (Fig. 6) were rod‑shaped and >10 µm long, underscoring CS‑FA’s role in modulating HCPT growth kinetics.
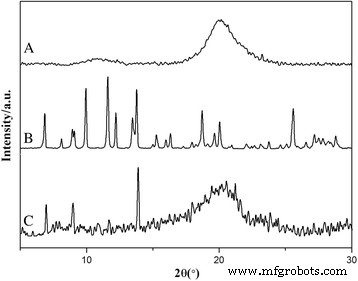
XRD patterns of (a) chitosan, (b) HCPT, and (c) HFNDs

SEM image of bulk HCPT crystals
In Vitro Drug Release Studies
Free HCPT released 30 % within 1 h and nearly 100 % by 18 h. HFNDs exhibited a markedly slower, sustained release over 48 h (Fig. 7), attributed to the CS‑FA shell’s diffusion barrier. This profile is ideal for maintaining therapeutic concentrations while minimizing peak‑related toxicity.
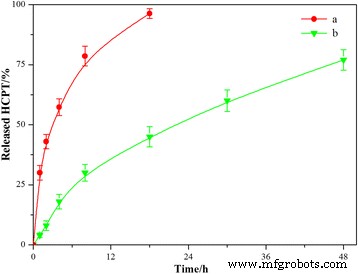
In vitro release profiles in PBS (pH 7.4) at 37 °C: (a) free HCPT; (b) HFNDs
Cellular Uptake
Confocal imaging (Fig. 8) revealed stronger HCPT fluorescence in HeLa cells treated with HFNDs versus non‑targeted NDs, indicating folate‑mediated uptake. Quantitative fluorescence (Fig. 9) corroborated a higher internalization rate for HFNDs, validating the targeting strategy.
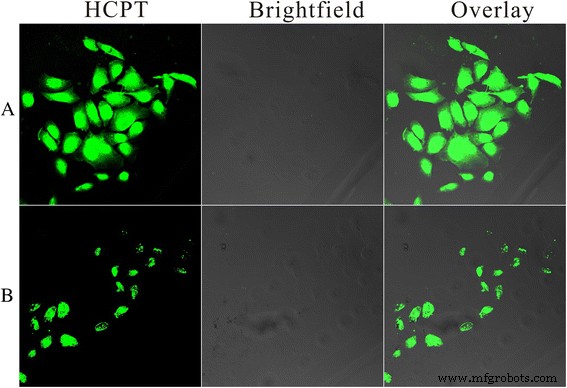
Intracellular HCPT delivery after 8 h: (a) HFNDs; (b) NDs
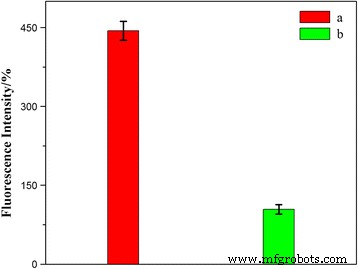
Fluorescence quantification of HeLa cells after 8 h: (a) HFNDs; (b) NDs; *p* < 0.05
Cytotoxicity Assays
MTT results (Fig. 10) showed that HFNDs achieved greater cell killing than free HCPT or NDs at equivalent HCPT concentrations, underscoring the dual benefit of targeted delivery and sustained release. The enhanced cytotoxicity is attributed to efficient receptor‑mediated internalization and a prolonged intracellular drug release profile.
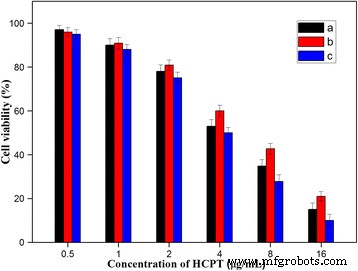
Cell viability after 24 h: (a) free HCPT; (b) NDs; (c) HFNDs; *p* < 0.05
Biodistribution
In vivo imaging (Fig. 11a) demonstrated negligible tumor signal for DiR‑NDs, whereas DiR‑HFNDs displayed a pronounced, time‑dependent accumulation, peaking at 12 h. Ex vivo analysis (Fig. 11b–c) confirmed higher tumor fluorescence for HFNDs, indicating successful folate‑mediated tumor targeting.
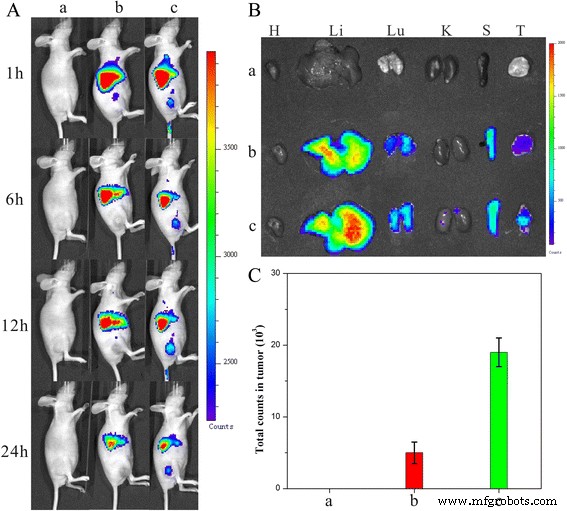
a Distribution and tumor accumulation; b ex vivo fluorescence; c DiR intensity in tumor; *p* < 0.05
Tumor Inhibition In Vivo
HFNDs achieved the most significant tumor growth suppression, with final tumor weights markedly lower than free HCPT or NDs (Fig. 12c). Histology (Fig. 12d) revealed extensive necrosis in HFND‑treated tumors. Importantly, HFNDs caused no weight loss or overt toxicity, contrasting with the pronounced side effects observed with free HCPT (Fig. 12b). These findings demonstrate that HFNDs combine potent antitumor activity with a favorable safety profile.
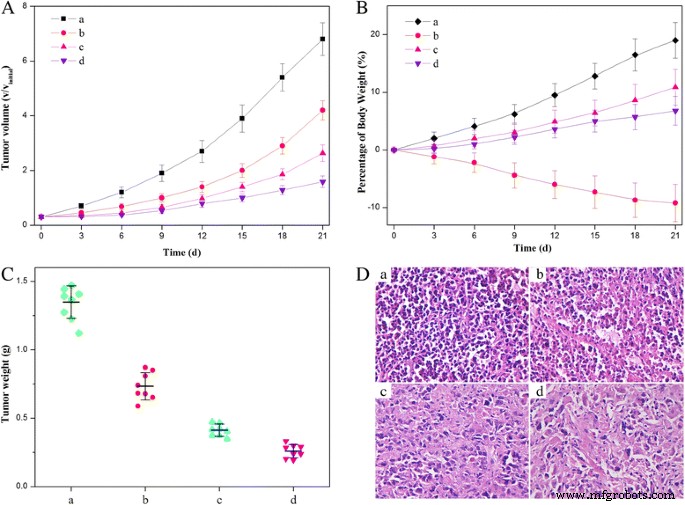
In vivo antitumor efficacy: (a) tumor volume; (b) body weight; (c) tumor weight; (d) H&E histology
Conclusions
We have introduced a fully green, solvent‑free route to produce folate‑functionalized HCPT nanoneedles that combine high drug loading, targeted cellular uptake, and sustained release. In vitro and in vivo data confirm superior anticancer activity and reduced systemic toxicity compared to conventional formulations. This platform exemplifies how environmentally responsible chemistry can be harnessed to advance nanoparticle‑based therapeutics.
Nanomaterials
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- Tecnoideal America Rebrands from TechnoMed Inc., Enhancing U.S. Presence in Medical Device Automation
- Targeted Folic Acid‑Conjugated Chitosan Nanoparticles Deliver Genistein, Boosting Anticancer Efficacy Against Cervical Cancer
- Methanol‑Enhanced PEDOT:PSS Hole Transport Layer Boosts Silicon/Organic Hybrid Solar Cell Efficiency to 12.2%
- S‑Doped Sb₂O₃ Nanocrystals: Tailored pH‑Controlled Visible‑Light Photocatalysts for Efficient Organic Degradation
- Reimagining Nanoparticle Therapies Beyond Cancer
- Designing Plasmonic Nanoparticle Strategies for Enhanced Organic Solar Cell Performance
- Targeted Co‑Delivery of 5‑Fluorouracil and LY294002 via PEG‑Lipid Nanoparticles Enhances Antitumor Efficacy in Esophageal Squamous Cell Carcinoma
- Redox‑Responsive Gelatin/Silica Nanogels Functionalized with AS1411 Aptamer for Targeted siRNA Delivery
- Pioneering Electron-Beam 3D Printing of Microgel Structures in Liquid Environments



