Yolk‑Shell Cu@C Nanocomposites: A High‑Performance Catalyst for Oxidative Carbonylation of Methanol to Dimethyl Carbonate
Abstract
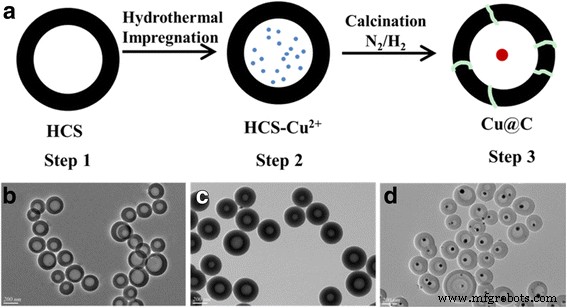
We present a simple, scalable route to produce yolk‑shell Cu@C nanocomposites in which copper cores (30–55 nm) are encapsulated inside hollow carbon spheres (≈210 nm diameter, 80 nm cavity). During pyrolysis, the confined cavity confines Cu nucleation, yielding monodisperse cores whose size is tuned by copper salt loading. By activating the carbon shell with KOH (mass ratio 1 : 4) we create a highly porous, micro‑/mesoporous network that dramatically enhances catalytic performance. The activated catalyst achieves a turnover frequency of 8.6 h⁻¹ and a methanol conversion of 17.1 % at 120 °C under 3 MPa CO/O₂ (2:1). Recyclability tests show negligible Cu leaching (<0.01 %) after seven cycles, underscoring the robustness of the yolk‑shell design for eco‑friendly dimethyl carbonate (DMC) synthesis.
Background
Dimethyl carbonate (DMC) is prized as a biodegradable, low‑toxicity solvent and intermediate for polycarbonates, isocyanates, and fuel additives. Its oxidative carbonylation from methanol—using CO, O₂, and MeOH—offers a high carbon‑utilization route with minimal environmental impact. Traditional catalysts rely on chlorine‑containing systems that suffer from corrosion, product contamination, and deactivation. Chlorine‑free copper and copper‑oxide catalysts on activated carbon have shown promise, yet agglomeration of Cu nanoparticles and loss of active species remain major challenges. Encapsulating Cu in protective shells, especially yolk‑shell nanostructures (YSNs), offers a solution: the shell shields the core while maintaining surface accessibility, providing a confined reaction environment that can improve activity and stability.
YSNs, particularly carbon‑shell variants, combine excellent electrical conductivity with chemical and thermal robustness. Recent work by Lu et al. demonstrated hollow spheres assembled via oleic‑acid templating and dihydroxybenzoic acid precursors. Building on this, we develop a ship‑in‑a‑bottle strategy to embed Cu cores within hollow carbon spheres (Cu@C), and subsequently tailor shell porosity through KOH activation.
Methods
Chemicals
All reagents were analytical grade: 2,4‑dihydroxybenzoic acid (DA), oleic acid, ammonia (25 %), formaldehyde, copper nitrate trihydrate Cu(NO₃)₂·3H₂O, potassium hydroxide (KOH), and methanol (MeOH). Deionized water was from a Milli‑Q system. O₂ (>99.99 %) and CO (>99.99 %) were supplied by Beijing ZG Special Gases.
Synthesis of Hollow Carbon Spheres (HCS)
Following Lu et al., 2.5 mmol DA and 7.5 mmol formaldehyde were dissolved in 95 mL deionized water. A 5 mL aqueous solution containing 56 µL oleic acid and 180 µL 25 % NH₃ was added at 30 °C with slow stirring for 30 min. The mixture was transferred to an autoclave and hydrothermally aged at 140 °C for 4 h. After centrifugation, washing with water and ethanol, the product was dried at 50 °C overnight and pyrolyzed at 700 °C for 2 h under N₂, yielding HCS.
Synthesis of Cu@C Nanocomposites
0.3 g HCS were dispersed in 30 mL copper nitrate solutions of varying concentration (0.03–0.24 M). The mixture underwent hydrothermal impregnation at 100 °C for 10 h, producing HCS‑Cu²⁺. Calcination at 400 °C for 2 h under 10 % H₂/90 % N₂ converted Cu²⁺ to Cu⁰, yielding Cu@C‑X (X = 0.03, 0.06, 0.12, 0.24).
KOH‑Activated Cu@A‑HCS Catalyst
HCS (0.3 g) were physically mixed with 0.15 g KOH (no water). The composite was heated to 700 °C at 10 °C min⁻¹ for 2 h under N₂. After repeated washing with dilute HCl and water to remove residual Cl⁻, the material was dried at 60 °C overnight. Cu impregnation was performed with 0.12 M Cu(NO₃)₂ under the same hydrothermal conditions, yielding Cu@A‑HCS.
Catalytic Evaluation
Oxidative carbonylation was conducted in a 25 mL stainless‑steel autoclave (Teflon‑lined) equipped with a magnetic stirrer. Each run used 0.2 g catalyst and 10 mL MeOH. After three CO purges, the reactor was pressurized to 3.0 MPa with CO/O₂ (2:1) at room temperature, then heated to 120 °C and stirred at 750 rpm for 1.5 h. Products were quantified by GC–FID. Recyclability was assessed by consecutive runs on the recovered catalyst.
The key reaction: 2 CH₃OH + ½ CO + O₂ → (CH₃O)₂CO + H₂O.
Quantification formulas:
- C_Cu (mmol g⁻¹) = (Cu wt % / 63.55) × 1000
- MeOH conversion (%) = (reacted MeOH / introduced MeOH) × 100
- DMC selectivity (%) = (2 × produced DMC / reacted MeOH) × 100
- TOF = produced DMC / (Cu mol × reaction time)
Results and Discussion
Textural and Thermal Properties of the Support
BET surface areas and pore volumes are summarized in Table 1. The initial HPS exhibits a low surface area (~23 m² g⁻¹), limiting conventional impregnation. Hydrothermal impregnation enhances copper precursor diffusion into the cavity. After impregnation and calcination, the BET surface area drops to 15 m² g⁻¹, confirming Cu loading inside the shell. TEM images (Fig. 2) confirm that Cu nanoparticles form exclusively within the hollow cavity.

Thermogravimetric analysis (Fig. 1b) shows a major weight loss near 215 °C, attributed to oleic‑acid decomposition and polymer carbonization. Calcination at 400 °C ensures complete carbonization while preventing Cu aggregation.
Structural Characterization of Cu@C Nanocomposites
For Cu@C‑0.12, TEM (Fig. 3) reveals uniform spheres (~210 nm) with an 80 nm cavity and Cu cores (~30 nm). HRTEM (Fig. 3b) shows lattice spacing of 0.18 nm, matching the Cu (200) plane. XRD (Fig. 3c) confirms face‑centered cubic Cu (JCPDS 04‑0836). Nitrogen sorption (Fig. 3d) yields a type I isotherm, a BET surface area of 365 m² g⁻¹, and a pore volume of 0.23 cm³ g⁻¹.
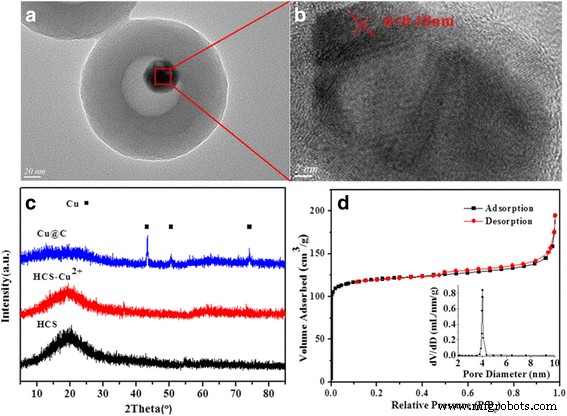
The confined nucleation–growth mechanism during pyrolysis explains the size tunability of Cu cores: higher Cu(NO₃)₂ loading leads to larger nuclei and subsequent growth within the cavity.
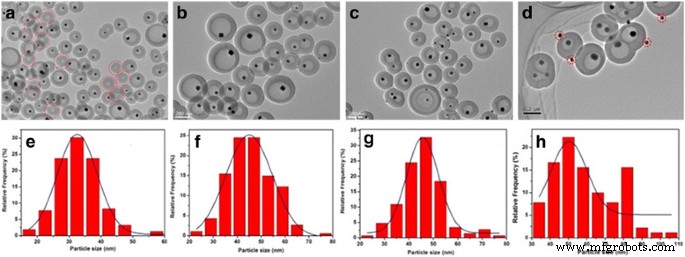
Size Control of Cu Cores
By varying Cu(NO₃)₂ concentration (0.03–0.24 M), Cu core sizes range from 30 ± 1.3 nm to 55 ± 2.5 nm (Fig. 4). TEM analysis of ≥150 particles per sample confirms this trend. At low Cu loading, some HCS remain unfilled; at high loading, minor Cu nanoparticles appear on the shell surface due to external precursor decomposition.
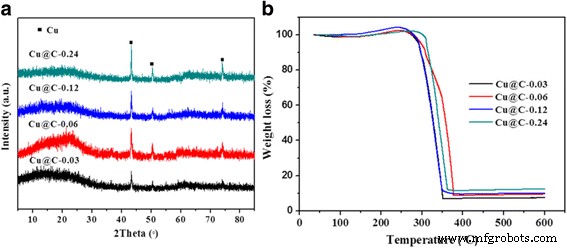
XRD (Fig. 5a) shows increasing peak intensity and sharpening with higher Cu loading, indicating larger crystallites (26.6–52.2 nm by Scherrer analysis). TG analysis (Fig. 5b) yields Cu loadings of 5.9–9.9 wt % (consistent with AAS).
Catalytic Performance of Cu@C‑X
Cu@C‑0.12, despite its high Cu content, delivers only 0.82 % MeOH conversion—attributable to limited shell porosity (0.23 cm³ g⁻¹, 365 m² g⁻¹). Enhancing shell porosity via KOH activation (1 : 4 KOH/HCS) creates a hierarchical micro/mesoporous network that dramatically improves mass transport.
Physicochemical Properties of Cu@A‑HCS
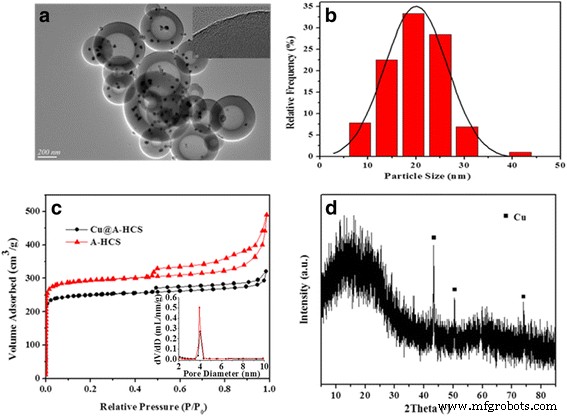
Activated Cu@A‑HCS (KOH/HCS = 1 : 4) retains the spherical morphology (Fig. 6a). Excessive KOH (>1 : 2) etches the spheres. TEM shows Cu nanoparticles (average 18 ± 2 nm) dispersed both inside the cavity and within the shell, while shell micro‑spores are evident. Nitrogen sorption (Fig. 6c) reveals a type IV isotherm with a BET surface area of 989 m² g⁻¹ (vs. 471 m² g⁻¹ for unactivated HCS) and increased mesopore volume, confirming successful activation. XRD (Fig. 6d) displays broadened Cu peaks, consistent with the ~15 nm particle size.
Catalytic Performance of Cu@A‑HCS
Cu@A‑HCS outperforms non‑activated Cu@C‑0.12 by nearly fourfold: TOF rises from 2.04 h⁻¹ to 8.6 h⁻¹, and MeOH conversion increases from 4.38 % to 17.1 % under identical conditions (Table 4). The enhanced activity stems from the higher surface area and mesoporosity, which facilitate reactant diffusion and concentration within the yolk cavity.
Recyclability studies show that Cu@A‑HCS retains >80 % of its initial activity after seven cycles, with Cu leaching <0.01 %. TEM and XRD of the recovered catalyst reveal negligible structural change, confirming the stability of the yolk‑shell architecture.
Conclusions
We have demonstrated a facile ship‑in‑a‑bottle strategy to produce yolk‑shell Cu@C nanocomposites with controllable core size. By tuning shell porosity through KOH activation, the catalyst achieves high TOF (8.6 h⁻¹) and methanol conversion (17.1 %) with remarkable stability and negligible leaching. This work highlights the potential of yolk‑shell architectures for green DMC production and offers a versatile platform for other catalytically relevant nanostructures.
Nanomaterials
- Nanocups: Advanced Light‑Bending Nanomaterials for Biomedical and Photonic Applications
- Electrospun TiO₂–Carbon Nanofiber Support Enhances PtRu Catalysts for Direct Methanol Fuel Cells
- Spherical Gold–Cockle Shell Calcium Carbonate Nanoparticles: Fabrication, Characterization, and Cytotoxicity for Biomedical Use
- High‑Performance CoFe/C Core–Shell Nanocomposites for Broadband Microwave Absorption
- Enhanced Photocatalytic Degradation of Rhodamine B Using SrTiO3/Bi5O7I Nanocomposites: Fabrication, Characterization, and Mechanistic Insights
- Platinum‑Based Catalysts on Carbon Supports and Conducting Polymers for Direct Methanol Fuel Cells: A Comprehensive Review
- Surfactant‑Free Cu₂O@Au Hollow Cubes Deliver Enhanced CO₂ Electroreduction to CO
- Efficient Fabrication of Mesostructured NR/SiO₂ Nanocomposites with Superior Thermal Stability and Hydrophobicity
- General Hydrothermal Fabrication of Carbon‑Coated Noble Metal Nanocomposites with Photothermal Function
- MoS₂ Nanocomposite-Based Enzyme-Free Glucose Biosensors: A Cutting-Edge Approach



