Optimized Biocompatibility of Anodic Tantalum Oxide Nanotube Arrays
Abstract
\nThis study investigates the biocompatibility of self‑organized TaOx nanotube arrays produced by electrochemical anodization, with diameters ranging from 20 to 90 nm. All as‑anodized samples were confirmed to be amorphous. Surface wettability varied with nanotube diameter, following Wenzel’s model of geometric roughness. In vitro tests with human fibroblasts revealed a clear wettability‑dependent response; the 35‑nm diameter arrays exhibited the highest biocompatibility, attributable to dense focal adhesion points and superior hydrophilicity. These findings demonstrate that tailoring nanotube diameter and surface roughness can enhance the biocompatibility of tantalum oxide surfaces.
\nBackground
\nTantalum (Ta) is a rare, hard, corrosion‑resistant, and bioinert metal whose ultrathin oxide film confers excellent biocompatibility. Its flexibility and low toxicity make it ideal for dental and orthopedic implants, bone reconstruction, and porous scaffolds that mimic native bone structure. Compared with titanium, tantalum supports greater extracellular matrix deposition, cellular adhesion, and proliferation. Recent studies have shown that nanostructured surfaces—especially self‑organized nanotube arrays—are pivotal in modulating cell behavior. Anodized Ta nanotubes have been reported to promote osseointegration more effectively than flat surfaces, and porous tantalum implants have achieved superior fixation in clinical trials. Building on prior work with TiO2 nanotubes, this study focuses on TaOx arrays of varying diameters, evaluating their influence on fibroblast adhesion and proliferation.
\nMethods
\nPreparation of TaOx Nanotubes
\nCommercial Ta sheets (0.127 mm, 99.7 % purity) were ultrasonically cleaned in acetone, isopropanol, ethanol, and water. Anodization was performed at 20 °C in 4.9 wt % HF sulfuric acid using a two‑electrode cell (Ta anode, Pt counter). Applied voltages (10–40 V) yielded nanotube diameters from 20 to 90 nm. Samples were exposed to low‑intensity UV light (≈2 mW cm–2) for 8 h before biocompatibility assays.
\nMaterial Characterization
\nScanning electron microscopy (SEM) quantified nanotube dimensions; X‑ray diffraction (XRD) and transmission electron microscopy (TEM) confirmed amorphous structure; energy‑dispersive spectroscopy (EDS) verified composition. Contact angles were measured with the extension method using water and culture medium.
\nHuman Fibroblast Cell Culture
\nMRC‑5 fibroblasts (BCRC No. 60023) were cultured in Eagle’s MEM with 10 % FBS, 2 mM L‑glutamine, 1.5 g L–1 NaHCO3, 0.1 mM non‑essential amino acids, and 1.0 mM pyruvate at 37 °C, 5 % CO2. Cells were seeded onto autoclaved TaOx sheets placed in 12‑well plates for subsequent assays.
\nCell Adhesion Assay
\nCells were seeded at 2.5 × 103 cells cm–2 and incubated for 3 days. After fixation, actin filaments were stained with rhodamine phalloidin and nuclei with DAPI. Fluorescence microscopy (AX80, Olympus) evaluated adhesion morphology. For SEM, cells were fixed in 2.5 % glutaraldehyde, dehydrated, critical‑point dried, and sputter‑coated with platinum.
\nCell Proliferation Assay
\nSeeding density of 1 × 104 cells cm–2 was used for a 1‑week culture. Proliferation was quantified with the WST‑1 reagent (Roche); absorbance was read at 450 nm (Spectral Max250).
\nStatistical Analysis
\nExperiments were performed in triplicate with at least three independent repeats. Data are expressed as mean ± SD and analyzed by one‑way ANOVA (SPSS 12.0). A p‑value < 0.05 was considered significant.
\nResults and Discussion
\nSEM images (Fig. 1) confirm well‑defined nanotube arrays with diameters proportional to anodization voltage: 20, 35, 65, and 90 nm. The 20‑nm tubes exhibit slightly irregular walls, likely due to lower electric field strength. Cross‑sectional SEM (Fig. 2) shows consistent tube lengths across diameters. XRD and TEM (Fig. 3) confirm that all arrays remain amorphous, which is advantageous for biocompatibility.
\n
SEM images of Ta foil and TaOx nanotubes with diameters of 20, 35, 65, and 90 nm.

Cross‑sectional SEM of TaOx nanotubes with diameters of 20, 35, 65, and 90 nm.
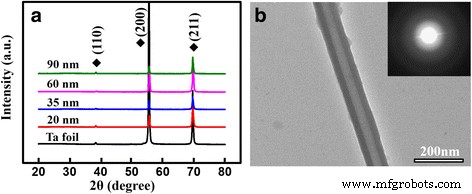
XRD spectra (a) and a TEM image of a 90‑nm TaOx nanotube (b).
Wettability measurements (Fig. 4) reveal that all arrays are highly hydrophilic, with contact angles decreasing as diameter approaches 35 nm, then increasing at 20 nm. This trend aligns with Wenzel’s model, where increased geometric roughness lowers contact angle on hydrophilic surfaces. The calculated roughness factor (Fig. 5) confirms that 35‑nm tubes possess the highest roughness, explaining their superior hydrophilicity.
\n
Water and culture medium droplets on Ta foil and TaOx arrays (20–90 nm). Contact angles are indicated.
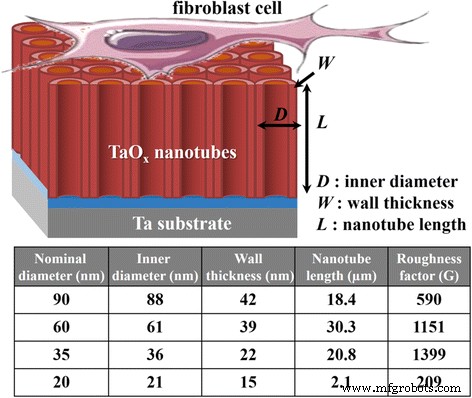
Schematic of nanotube geometry and calculated roughness factors for each diameter.
Fluorescence microscopy (Fig. 6) and SEM (Fig. 7) demonstrate that fibroblasts adhere and spread most effectively on 35‑nm tubes, forming robust actin networks and focal adhesions. The 20‑nm arrays, while offering more potential focal points, exhibit reduced hydrophilicity, impairing adhesion. Cell coverage area, quantified with ImageJ, mirrors the contact‑angle trend: maximal at 35 nm, lower at 20 nm and 90 nm.
\n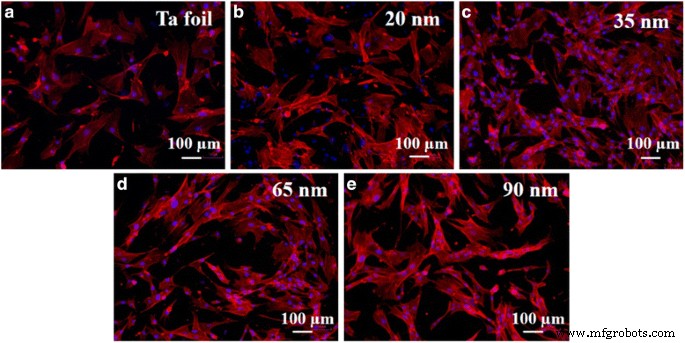
Fluorescence images of fibroblast actin (red) and nuclei (blue) on Ta foil and TaOx arrays (20–90 nm).

SEM images of fibroblast adhesion and proliferation on Ta foil and TaOx arrays (20–90 nm). Coverage areas are marked.
The WST‑1 assay (Fig. 8) confirms that proliferation peaks on 35‑nm tubes, matching the wettability and adhesion data. No significant difference is observed between the flat Ta foil and TaOx arrays overall, underscoring the importance of nanoscale geometry and surface energy in cell response.
\n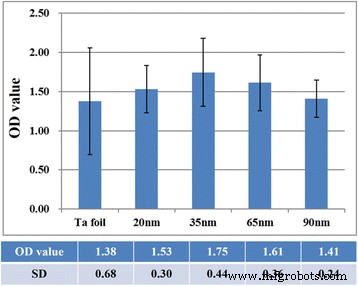
Optical densities from WST‑1 assay for fibroblasts on Ta foil and TaOx arrays (20–90 nm). Error bars represent SD.
Conclusions
\nSelf‑organized TaOx nanotube arrays with diameters from 20 to 90 nm were fabricated via anodization and characterized as predominantly amorphous. Wettability varied systematically with diameter, following Wenzel’s roughness model. In vitro fibroblast studies revealed a clear dependence on surface hydrophilicity and nanotube geometry; the 35‑nm arrays provided optimal focal adhesion density and cell coverage, resulting in the highest biocompatibility. These results demonstrate that tailoring TaOx nanotube diameter and roughness can substantially improve the biological performance of tantalum‑based implants.
\nNanomaterials
- Enhanced Retention in TaO/HfO and TaO/AlO RRAM: A Self‑Rectifying Switching Model
- Enhanced Photocatalytic Water Splitting with Au‑Plasmonic Nanoparticles on TiO₂/MoO₃ Nanotube Photoelectrodes
- Optimizing PdAu/VGCNF Anode Catalyst for Enhanced Glycerol Fuel Cell Performance
- Facile Thermal Decomposition Loading of CeO₂ Nanoparticles onto Anodic TiO₂ Nanotube Arrays
- Titania Nanotube Arrays via Electrochemical Anodization: Synthesis, Modifications, and Biomedical Applications
- High‑Sensitivity Fe³⁺ Detection Using Ag‑Functionalized TiO₂ Nanotube Arrays via Anodic Stripping Voltammetry
- Crystal Structure Drives Supercapacitive Performance of Hydrogenated TiO₂ Nanotube Arrays
- Boosting ZnO UV Emission with Silver Nanoparticle Arrays via Surface Plasmon Resonance
- C Arrays Explained: Efficient Data Storage & Access
- Mastering Arrays in C#: Efficient Data Storage & Access



