64Cu‑Labeled AGuIX Nanoprobes: PET‑Guided Radiotherapy and Radiosensitization in HepG2 Tumor‑Bearing Mice
Abstract
Radiation therapy for liver cancer is constrained by the liver’s low tolerance to ionising radiation, making radiosensitizers essential to lower required doses. AGuIX nanoparticles—ultra‑small, gadolinium‑based carriers—are uniquely suited to deliver radioisotopes or fluorophores for SPECT, PET, fluorescence imaging, and multimodality imaging, while their high atomic number also confers tumour‑sensitising properties. Understanding their biodistribution and pharmacokinetics is therefore critical to quantify tumour retention during radiotherapy. In this study, we efficiently labelled AGuIX with 64Cu via its intrinsic DOTA chelator. Biodistribution data revealed pronounced accumulation of 64Cu‑AGuIX in HepG2 xenografts in nude mice, indicating its promise as a theranostic probe for image‑guided radiotherapy in hepatocellular carcinoma (HCC). Transmission electron microscopy confirmed nanoparticle uptake within HepG2 cells. Radiation experiments showed a significant reduction in 18F‑FDG uptake in tumours irradiated 1 h post‑AGuIX injection, providing proof‑of‑concept that AGuIX functions as a PET‑visible radiosensitizer for liver cancer radiotherapy.
Background
Hepatocellular carcinoma (HCC) ranks among the world’s most common malignancies, with 782,500 new cases and 745,500 deaths reported in 2012, of which 70–90 % were HCC. Most patients present at an advanced stage, limiting curative options to 20–25 % of cases. Consequently, radiotherapy has become a key component of multidisciplinary HCC management. However, normal liver tissue limits dose escalation, as radiation‑induced liver disease (RILD) poses a severe risk. Radiosensitizers that preferentially accumulate in tumour tissue can increase tumour cell kill at lower doses, thereby mitigating normal‑tissue toxicity.
AGuIX, a novel gadolinium‑based nanoparticle (~5 nm diameter) first described in 2013, is rapidly renally cleared and can be functionalised with radioactive or fluorescent labels for SPECT, PET, MRI, or fluorescence imaging. The high gadolinium content endows AGuIX with tumour‑sensitising capabilities, demonstrated in vitro across diverse cell lines with sensitisation enhancement ratios (SER) ranging from 1.1 to 2.5. Because AGuIX shows low hepatic background yet high tumour uptake—thanks to the enhanced permeability and retention (EPR) effect—it is an attractive candidate for HCC radiosensitisation.
Despite extensive preclinical work on AGuIX for MRI‑guided radiotherapy, its pharmacokinetics remain incompletely characterised. Precise biodistribution data are essential to optimise dosing schedules and predict therapeutic windows. The positron emitter 64Cu (T1/2 = 12.4 h) offers the temporal flexibility to image both rapid and slow‑clearing agents. Here we report the first in vivo biodistribution and PET imaging of 64Cu‑labeled AGuIX in HepG2 tumour‑bearing nude mice, and we evaluate its radiosensitising effect using 18F‑FDG PET/CT.
Methods
General Information
AGuIX nanoparticles (sub‑5 nm, dehydrated, spherical) were sourced from Nano‑H (Lyon, France) and rehydrated in sterile DEPC‑treated water. Human HCC HepG2 cells (ATCC) were cultured in MEM supplemented with 10 % FCS. The 64Cu isotope was obtained from Wisconsin University. Male BALB/c athymic nude mice (6 weeks, 16–18 g) were used, with all protocols approved by the Institutional Animal Care and Use Committee at the University of Virginia.
Transmission Electron Microscopy (TEM)
AGuIX (0.5 mM) was incubated with HepG2 cells for 1 h, followed by washing with 0.1 M PBS and centrifugation. Cell pellets were fixed in 4 % formaldehyde and 1 % glutaraldehyde for TEM imaging.
64Cu Radiolabeling
AGuIX (10 µmol) was mixed with 100 µl 0.5 M NH4OAc (pH 5.5) and incubated for 5 min. 1–3 mCi of 64CuCl2 (0.1 N HCl) was added, and the mixture was heated at 37 °C for 1 h. The product was purified by 3 kDa Amicon filtration. Radiochemical purity was confirmed by iTLC (20 mM citric acid) and consistently exceeded 98 %. Specific activity ranged from 3–10 MBq µmol–1, with final yields of 50–100 MBq per synthesis.
Tumor Models
HepG2 cells (5 × 106) were resuspended in 0.1 ml HBSS and injected subcutaneously into the right flank of each nude mouse. Tumours became palpable after ~2 weeks.
Biodistribution of 64Cu‑AGuIX
Ten mice (5 male, 4 female) received intraperitoneal injections of ~0.9 MBq 64Cu‑AGuIX (0.2 ml). At 9, 21, and 40 h post‑injection, mice were euthanised, and organs (heart, muscle, lung, kidney, spleen, liver, tumour) were harvested, weighed, and counted in a γ‑counter. Data were expressed as % injected dose per gram (% ID/g).
Micro‑PET Imaging
Each tumour‑bearing mouse received 22.2 MBq 64Cu‑AGuIX intraperitoneally. PET scans were acquired at 9 h and 21 h under isoflurane anaesthesia, using a SuperArgus system (Sedecal, Spain).
Irradiation and 18F‑FDG PET/CT
Twelve mice were divided into three groups (n = 4). Baseline PET imaging was performed 30 min after tail‑vein injection of 16.4 ± 4.7 MBq 18F‑FDG. Groups received either saline, 1 mg, or 10 mg AGuIX intraperitoneally 1 h before irradiation. Irradiation was delivered at 250 kV, 8 mA, 2‑mm Al filter (1.2 Gy min–1) for a total of 6 Gy; the protocol was repeated the following day. 24 h after the second dose, a second 18F‑FDG PET scan (11.1 ± 1.0 MBq) was performed. Standardised uptake values (SUVmax) were measured in tumours and normalised to liver SUVave to obtain T/L ratios.
Statistical Analysis
All experiments were performed in triplicate. Data are presented as mean ± SE. Significance was assessed by two‑tailed unpaired t tests or one‑way ANOVA (p < 0.05 considered significant).
Results and Discussion
While contrast‑enhanced MRI can detect AGuIX, PET offers superior sensitivity and quantitative accuracy, enabling detection of nanomolar concentrations that are below MRI’s threshold. Here we demonstrate that 64Cu‑labeled AGuIX provides clear tumour delineation and prolonged retention in HepG2 xenografts, supporting its use for PET‑guided radiotherapy.
TEM Study
Incubation of HepG2 cells with 0.5 mM AGuIX for 1 h resulted in cytoplasmic uptake, as visualised at ×6 500 and ×52 000 magnification (Figure 1). The nanoparticles maintained a well‑dispersed morphology, indicating cellular stability and compatibility.
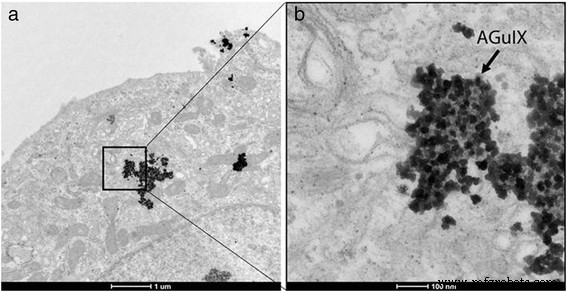
Localization of AGuIX within HepG2 cells. a. TEM images (× 6500) depicting uptake. b. Magnified view (× 52000) showing cytoplasmic distribution.
Radiolabeling
One‑step chelation of 64Cu to AGuIX via the built‑in DOTA achieved >98 % radiochemical yield. The resulting specific activity was 3–10 MBq µmol–1, with final product activity of 50–100 MBq per synthesis.
Biodistribution Studies
After intraperitoneal injection, 64Cu‑AGuIX exhibited robust tumour uptake (7.82 ± 1.50, 8.43 ± 6.23, 6.84 ± 1.40 % ID/g at 9, 21, and 40 h, respectively). Non‑target organs retained <1 % ID/g, confirming rapid clearance. Kidney uptake was notably lower (~5 % ID/g) than reported for intravenous administration, likely due to continuous peritoneal absorption. These data support the use of 64Cu‑AGuIX as a quantitative tool to guide radiosensitiser dosing.
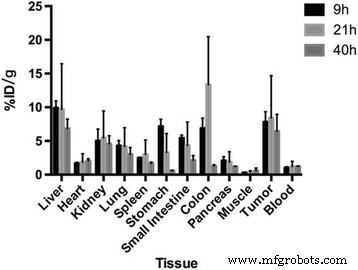
Biodistribution of 64Cu‑AGuIX in tumour‑bearing mice (% ID/g at 9, 21, and 40 h). Mean ± SD, n = 3.
Micro‑PET Imaging
Clear tumour signal was observed at 9 h, with even higher contrast at 21 h as background activity declined (Figure 3). The data corroborate the biodistribution findings and demonstrate the practical feasibility of PET‑guided treatment planning.
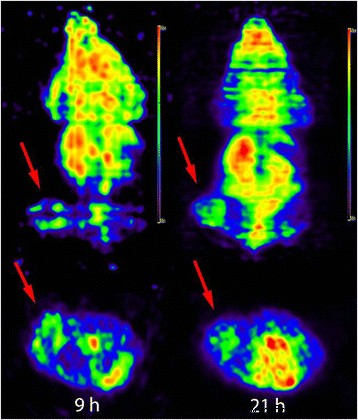
Micro‑PET images of tumour‑bearing mice. Upper panel (coronal view) and lower panel (transverse view) at 9 h and 21 h post‑injection. Tumour indicated by red arrow.
18F‑FDG PET/CT Evaluation of Irradiated Xenografts
All irradiated mice displayed reduced tumour 18F‑FDG uptake. SUVmax ratios (B/A) were 1.03 ± 0.03 (saline), 1.04 ± 0.04 (1 mg AGuIX), and 1.24 ± 0.02 (10 mg AGuIX). The 10 mg group showed a statistically significant increase in tumour‑to‑liver uptake ratio compared to the 1 mg and saline groups (p < 0.001), confirming dose‑dependent radiosensitisation. No difference was observed between 1 mg AGuIX and saline, indicating that higher doses are required for clinical benefit.
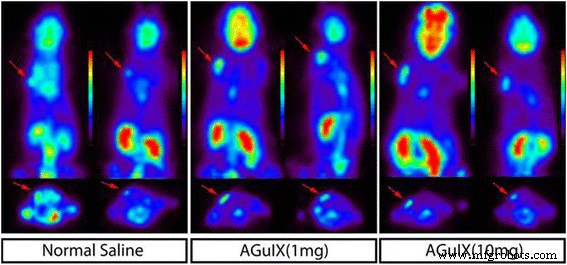
Pre‑ and post‑irradiation 18F‑FDG PET images for saline (left), 1 mg AGuIX (middle), and 10 mg AGuIX (right). Images share identical colour scales.
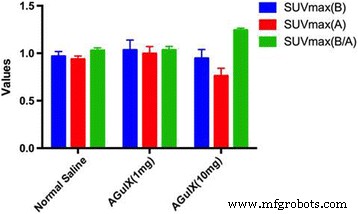
Quantitative analysis: T/L (B) – tumour to liver SUVave before irradiation; T/L (A) – after irradiation; T/L (B/A) – ratio of the two. 1 mg and 10 mg AGuIX groups shown.
While renal uptake of radiolabelled AGuIX can be high after intravenous injection, intraperitoneal delivery mitigates this exposure, resulting in sustained tumour retention with minimal kidney exposure. Future dosimetry studies will clarify organ‑specific radiation tolerance and support clinical translation.
Conclusions
We have successfully radiolabelled AGuIX with 64Cu, achieving >98 % purity and substantial tumour uptake that persists for >40 h in HepG2 xenografts. PET imaging demonstrates clear tumour delineation and provides a quantitative readout for treatment planning. Radiosensitisation studies using 18F‑FDG PET confirm that a 10 mg intraperitoneal dose of AGuIX enhances the therapeutic effect of 6 Gy radiotherapy in HCC models. These findings validate AGuIX as a dual‑function theranostic nanoprobe for image‑guided radiotherapy and highlight the need for comprehensive dosimetry before clinical application.
Nanomaterials
- Boron Carbon Oxynitride: A Novel Metal‑Free Photocatalyst with Enhanced Solar‑Driven Hydrogen Production and Dye Degradation
- Nanoparticle-Enhanced Wormlike Micellar System: Design, Rheology, and Mechanistic Insights
- Enhanced Visible‑Light Photocatalytic Degradation of Rhodamine B Using Bi<sub>4</sub>Ti<sub>3</sub>O<sub>12</sub>/Ag<sub>3</sub>PO<sub>4</sub> Heterojunction Nanocomposites
- Enhanced Photocatalytic Degradation of Rhodamine B Using SrTiO3/Bi5O7I Nanocomposites: Fabrication, Characterization, and Mechanistic Insights
- High‑Efficiency Tumor‑Targeted Photo‑Chemo Theranostic Nanoparticles: Artesunate‑Loaded, ICG‑Conjugated Human Serum Albumin
- Wireless Magnetoelastic Nanobiosensor Achieves Ultra‑Sensitive Atrazine Detection
- Double‑Gated Nanohelix as a Tunable Binary Superlattice: Band Engineering and Optoelectronic Prospects
- Targeted Doxorubicin Delivery Using Anti-EpCAM Aptamer-Functionalized Liposomes: Efficacy in C26 Colon Carcinoma Models
- Emodin‑Loaded Stearic Acid‑G‑Chitosan Oligosaccharide Nanomicelles: Design, Characterization, and Antitumor Activity
- Advances in Thermosensitive Hydrogels for Targeted Tumor Therapy



