Ultra‑sensitive Amperometric Biosensor for Matrix Metalloproteinase‑7 Detection Using Pd‑Functionalized Carbon Nanocomposites
Abstract
Matrix metalloproteinase‑7 (MMP‑7) drives tumour invasion by cleaving extracellular matrix and peptide bonds between alanine and leucine. Here we present a Pd‑functionalized carbon nanocomposite (Pd‑CNC) that acts as a high‑impedance enhancer in an amperometric MMP‑7 biosensor. Palladium nanoparticles catalyze the oxidation of 4‑chloro‑1‑naphthol (4‑CN) by H₂O₂, producing an insoluble, high‑resistance precipitate on the electrode. The inherently poorly conductive carbon nanospheres further increase resistivity, amplifying the current differential between the analyte‑free and analyte‑present states. The sensor achieves a detection range of 100 fg mL⁻¹ to 100 ng mL⁻¹ and a limit of detection (LOD) of 17.38 fg mL⁻¹.
Background
MMP‑7, a zinc‑dependent endopeptidase, is central to tumour progression and metastasis due to its ability to degrade extracellular matrix components [1]. Elevated serum MMP‑7 levels correlate with lymph node metastasis in salivary gland cancer, colon adenocarcinoma, and high‑grade renal cell carcinoma [2–6]. Accurate, ultrasensitive detection of MMP‑7 is therefore a priority for early cancer diagnosis [7]. Conventional colorimetric, electrochemiluminescent, and FRET assays typically reach only the picogram range, whereas electrochemical biosensors can attain femtogram‑level LODs [8–11]. However, many electrochemical protocols rely on enzymatic catalysts that suffer from environmental instability and low durability [12–19]. Noble metals such as Pd exhibit superior catalytic activity and chemical robustness, and when combined with carbon supports they provide a stable, high‑surface‑area platform for bioanalytical applications [20–23].
In this work we engineer a Pd‑functionalized carbon nanocomposite that serves as an impedance enhancer, leveraging two synergistic effects: (1) the poor electrical conductivity of carbon nanospheres, and (2) Pd‑catalyzed formation of a resistive precipitate from 4‑CN/H₂O₂ oxidation. This design dramatically lowers the biosensor’s LOD to 17.38 fg mL⁻¹, demonstrating its potential for clinical MMP‑7 monitoring.
Methods
Materials
All reagents were analytical grade. HAuCl₄·3H₂O, H₂PtCl₄, 4‑CN, glucose, H₂O₂ (30 %), bovine thrombin, graphene oxide (GO), BSA, peptide (NH₂‑KKKRPLALWRSCCC‑SH), NSE, PSA, MMP‑2, MMP‑7, and clinical serum samples were sourced from commercial suppliers. Ultrapure water (18 MΩ cm) and 0.1 M PBS (pH 7.4) were used throughout.
Apparatus
Microwave synthesis employed a CEM Discover® SP reactor. Morphology and elemental analysis were conducted via HITACHI S‑4800 SEM, H7650 TEM, and SU8010 SEM/EDS. Electrochemical measurements used a CHI600 workstation with a three‑electrode cell (4 mm GCE, Pt counter, Ag/AgCl reference). High‑resolution TEM was performed on a Tecnai G²F30 (300 kV).
Synthesis of Pd‑Functionalized Carbon Nanocomposites
Glucose (4 g) was dissolved in 40 mL ultrapure water, then hydrothermally treated at 170 °C for 5 h to yield carbon nanospheres. The suspension was centrifuged, washed, and redispersed in 8 mL water. To functionalize with Pd, 1 mL of the nanosphere suspension was mixed with 25 µL of 1 mM H₂PdCl₄ and microwaved at 100 °C, 250 W for 15 min. The resulting Pd‑carbon nanospheres were purified by centrifugation and redispersed in 1 mL water. BSA (1 wt %) was then incubated with the Pd‑nanospheres for 1 h to block nonspecific binding, followed by washing and storage at 4 °C.
Electrodeposition of Au‑rGO on GCE
The GCE was polished with 0.05 µm alumina, sonicated, and rinsed. GO (8 mg) was dispersed in 20 mL water by 2 h sonication, then 200 µL 4 wt % HAuCl₄ was added. Au‑reduced GO (Au‑rGO) was electrodeposited by cyclic voltammetry from +1.5 to –1.5 V (50 mV s⁻¹). The modified electrode was washed and dried.
Biosensor Fabrication
The Au‑rGO/GCE was incubated with 40 µL (50 µM) peptide for 40 min at 37 °C, then cross‑linked with 50 µL (0.10 wt %) glutaraldehyde for 30 min. Finally, 20 µL of Pd‑CNC suspension was dropped onto the peptide‑modified electrode and allowed to rest for 1 h. Each step was followed by thorough rinsing with ultrapure water.
Electrochemical Measurement
After incubation with 1 ng mL⁻¹ MMP‑7 for 1 h at 37 °C, the electrode was rinsed and 50 µL of 1 mM 4‑CN containing 10 mM H₂O₂ was added to initiate the precipitation reaction for 50 min. Square‑wave voltammetry (SWV) was performed from –0.2 to 0.6 V (5 mM [Fe(CN)₆]³⁻/⁴⁻, 0.1 M PBS, pH 7.4) with a 25 mV pulse amplitude and 4 mV s⁻¹ step.
Results and Discussion
Principle of the Peptide‑Cleavage Biosensor
As illustrated in Scheme 1, the Au‑rGO/GCE provides a highly conductive platform that supports peptide immobilization via Au‑S bonds. Pd‑CNCs act as a catalytic impedance enhancer; upon addition of 4‑CN/H₂O₂, Pd catalyzes the oxidation of 4‑CN, generating an insoluble precipitate that significantly increases electrode resistance. When MMP‑7 cleaves the peptide (between Ala and Leu), Pd‑CNCs are partially removed, reducing the resistive layer and producing a measurable current increase. This dual amplification—peptide cleavage and catalytic precipitation—drives the sensor’s ultrasensitivity.
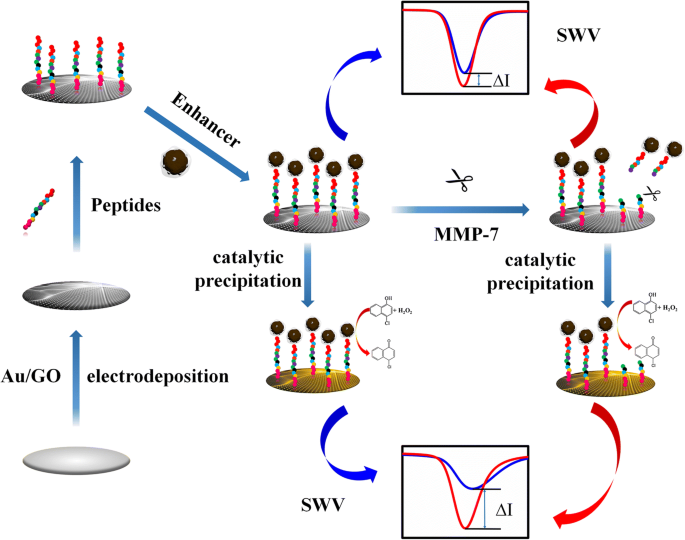
Schematic illustration of the Pd‑functionalized carbon nanosphere as an impedance enhancer for amperometric assay of MMP‑7
SEM imaging of the Pd‑CNC/peptide/Au‑rGO/GCE after the precipitation step (Fig. 1a) shows a uniform, insoluble layer, confirming successful formation of the high‑resistance precipitate. The sensor therefore achieves a markedly improved signal‑to‑noise ratio.

SEM images of Pd‑CNC/peptide/Au‑rGO/GCE (a) treated with catalytic precipitation reaction (b)
Characterisation of the Pd‑CNCs
Transmission electron microscopy (TEM) confirms uniform Pd nanoparticle deposition on 150 nm carbon nanospheres (Fig. 2a–c). EDS spectra (Fig. 2d–f) validate the presence of C, O, Pd, and BSA‑derived S/N, indicating successful functionalization. High‑resolution TEM (Fig. 3a–d) reveals face‑centered cubic Pd crystallites with an observable (1,1,1) lattice plane.
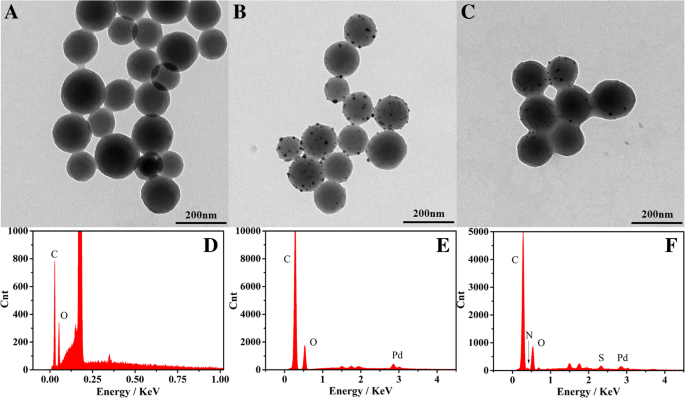
TEM micrographs of carbon nanosphere (a), Pd‑carbon nanospheres (b) and Pd‑CNCs (c) EDS of carbon nanosphere (d), Pd‑carbon nanospheres (e) and Pd‑CNCs (f)
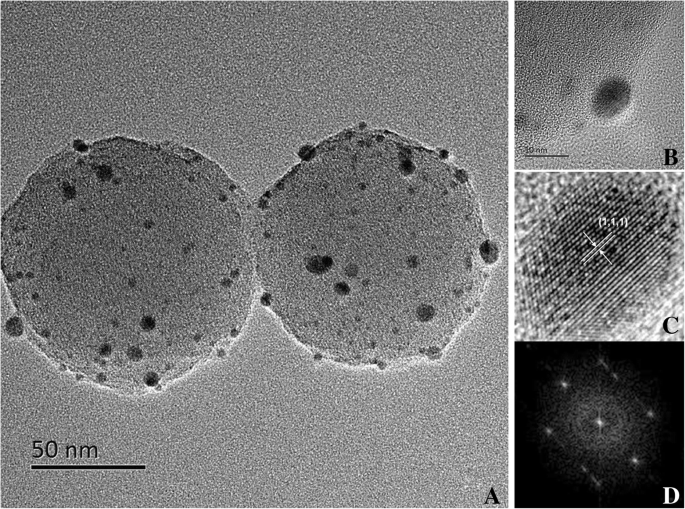
HR‑TEM images of Pd‑carbon nanospheres (a), magnified images of Pd nanoparticles (b, c) and FFT images of Pd nanoparticles (d)
Construction Monitoring by SWV and EIS
Figure 4a displays the SWV evolution during electrode modification. The peak current rises to ~420 µA after Au‑rGO deposition, then decreases upon peptide and Pd‑CNC attachment due to added impedance. Cleavage by MMP‑7 restores the current (~48.7 µA ΔI₁). Following the precipitation step, the current drops to 136.1 µA (ΔI₂ = 81.2 µA). EIS (Fig. 4b) corroborates these changes: the semicircle diameter increases with each modification, and the resistance falls after peptide cleavage, reflecting the removal of Pd‑CNCs.
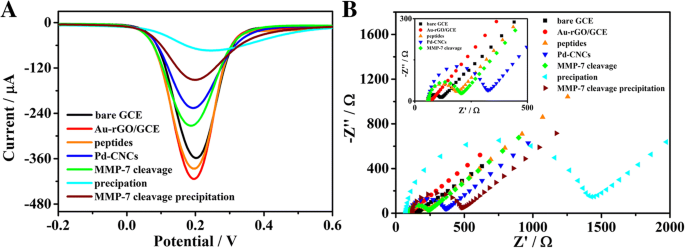
SWV (a) and EIS (b) responses of modification process of electrode in 5 mM [Fe(CN)₆]³⁻/⁴⁻ phosphate buffered (0.1 M, pH 7.4)
Optimization of Detection Conditions
Optimal peptide concentration was found at 50 µM; incubation time of 40 min maximized signal stability. Peptide cleavage and precipitation times of 50 min each yielded plateaued responses, ensuring complete reactions. These conditions were adopted for subsequent analyses.
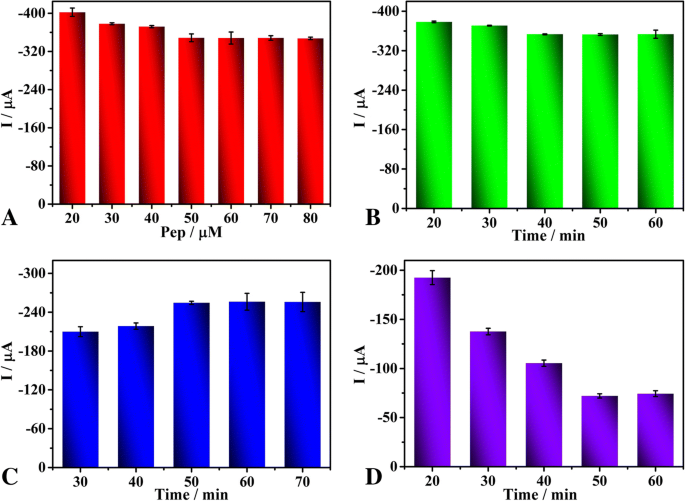
Effects of peptide concentration (a), incubation time of 50 µM peptide (b), peptide cleavage time (c) and precipitation reaction time (d) on the current responses of the biosensor
Analytical Performance
The biosensor exhibits a linear response (R² = 0.9967) between current peak and log[MMP‑7] over 0.1 pg mL⁻¹–100 ng mL⁻¹ (Eq. 1). The LOD, calculated at S/N = 3, is 17.38 fg mL⁻¹. Compared with recent peptide‑cleavage sensors, this device outperforms others in sensitivity and dynamic range (Table 1).
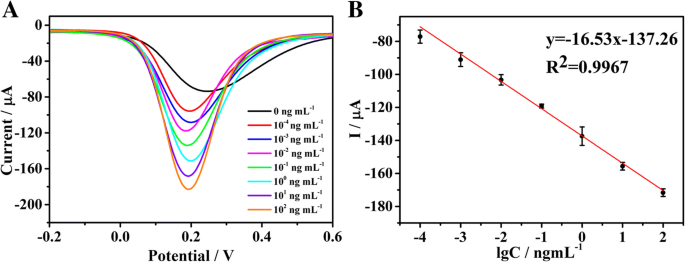
a SWV responses of electrochemical detection for MMP‑7 in 5 mM [Fe(CN)₆]³⁻/⁴⁻ phosphate buffered (0.1 M, pH 7.4) at concentrations from 100 fg mL⁻¹ to 100 ng mL⁻¹. b Linear calibration curve of current and the logarithm of the concentration of MMP‑7. Error bars are standard deviations for n = 3.
Specificity, Stability and Practical Application
Reproducibility tests (n = 3) yielded relative standard deviations of 1.3–4.0 % across a 0.01–100 ng mL⁻¹ range. Interference studies with MMP‑2, NSE, PSA, thrombin, and BSA (100 ng mL⁻¹ each) showed negligible signal change, confirming high specificity. Storage at 4 °C for 28 days produced RSD < 10 %, indicating robust stability. Recovery experiments in spiked serum (10–100 ng mL⁻¹) returned 86.3–117.2 %, demonstrating clinical feasibility.
Conclusions
We have engineered a Pd‑functionalized carbon nanocomposite that serves as an impedance enhancer, harnessing Pd’s catalytic prowess for 4‑CN oxidation and the poor conductivity of carbon nanospheres to create a highly resistive, precipitate‑based signal amplification. The resulting amperometric biosensor delivers a LOD of 17.38 fg mL⁻¹ and a wide linear range (0.1 pg–100 ng mL⁻¹) for MMP‑7, coupled with excellent specificity, reproducibility, and stability. This platform illustrates the power of impedance enhancers in electrochemical assays and paves the way for next‑generation biosensors in oncology diagnostics.
Abbreviations
- 4‑CN:
4‑chloro‑1‑naphthol
- BSA:
Bovine serum albumin
- EDS:
Energy dispersive X‑ray spectroscopy
- FCC:
Face‑center cubic
- FFT:
Fast Fourier transformation
- GCE:
Glassy carbon electrode
- GO:
Graphene oxide
- HR‑TEM:
High‑resolution transmission electron microscope
- LOD:
Limit of detection
- MMP‑2:
Matrix metalloproteinase‑2
- MMP‑7:
Matrix metalloproteinase‑7
- NSE:
Neuron‑specific enolase
- PBS:
Phosphate buffer solution
- Pd‑CNCs:
Pd‑functionalized carbon nanocomposites
- PSA:
Prostate‑specific antigen
- RSD:
Relative standard deviations
- SEM:
Scanning electron microscope
- SWV:
Square wave voltammetry
- TBS:
Thrombin of bovine serum
- TEM:
Transmission electron microscope
Nanomaterials
- McLaren 720S Enhanced: 3D‑Printed Carbon‑Fiber Body Lightens Car by 9%
- Enhancing Lithium‑Ion Battery Cathodes: LiNi0.8Co0.15Al0.05O2/Carbon Nanotube Composite with Superior Electrochemical Performance
- Visible‑Light‑Assisted Au Nanoparticle‑Modified Glassy Carbon Electrodes for Sensitive Uric Acid Detection
- Synergistic Reinforcement of Cu/Ti₃SiC₂/C Nanocomposites with Graphene and MWCNTs: Microstructure and Mechanical Performance
- Si‑Rich Alumina Supports Enhance Vertical SWCNT Growth and Nanofiltration Membrane Selectivity
- Efficient Synthesis of CNT–Cu₂O Nanocomposites for Rapid p‑Nitrophenol Reduction: A Low‑Cost, Reusable Catalyst
- ZnSe/N‑Doped Carbon Composites Derived from ZIF‑8 Deliver High Capacity and Long‑Term Cycling for Lithium‑Ion Batteries
- Efficient Fabrication of Mesostructured NR/SiO₂ Nanocomposites with Superior Thermal Stability and Hydrophobicity
- Affordable Green Fluorescent Carbon Dots: Enhanced Bioimaging & Targeted Drug Delivery
- Stable Carbon‑Dot Frameworks with Superior Antibacterial Efficacy



