Synthetic Rubies: Production Methods, History, and Industrial Applications
Background
Diamonds, rubies, sapphires, and emeralds are celebrated for their beauty and rarity. While the diamond is the most coveted, the ruby ranks as the second‑hardest gemstone, boasting exceptional resistance to acids and other harsh chemicals. Due to its scarcity, a high‑quality ruby can be worth up to four times a similarly sized diamond.
Both rubies and sapphires are crystalline forms of aluminum oxide (corundum). Their color derives from trace minerals: chromium imparts the characteristic red hue to rubies, whereas sapphire contains no chromium and appears in a spectrum of colors from blue to colorless.
Natural rubies are sourced from a handful of global deposits—Myanmar, Thailand, Sri Lanka, Afghanistan, Tanzania, and North Carolina. While translucent and transparent stones are prized for jewelry, opaque or translucent crystals find use in ornamental items such as clock bases.
Beyond aesthetics, rubies serve diverse industrial roles. Their extreme hardness makes them ideal for durable thread guides in textile machinery. Even harder than steel, ruby bearings are critical in precision devices like watches, compasses, and electrical meters. Their broad optical transparency—from ultraviolet to infrared—renders them indispensable in lasers and masers.
To meet industrial demands for flawless crystals of precise sizes and shapes, synthetic rubies are produced. With minimal impurities, synthetic gems replicate the chemical, physical, and optical properties of their natural counterparts. Roughly 75 % of modern synthetic ruby production is directed toward industrial applications.
History
Rubies have been mined for over 8,000 years. Ancient cultures attributed mystical powers to the stone: the Greeks believed rubies could melt wax; Hindus thought they could boil water; Burmese and Native American traditions held that rubies offered protection because of their blood‑red color.
The quest to create rubies began early. French chemist Marc A. Gaudin produced the first synthetic rubies in 1837, but they became opaque upon cooling, rendering them gem‑unsuitable. After decades of experimentation, he abandoned the effort.
In 1885, several gemstones sold as rubies were discovered to be manmade—later identified as “Geneva rubies.” Their creation involved melting powdered aluminum oxide and a small amount of chromium oxide using torches, a technique that would evolve into the flame fusion method.
In 1877, chemist Edmond Frémy and a student melted a solution of aluminum oxide in lead oxide over 20 days, producing minute ruby crystals that were too small and costly for jewelry. A few years later, Frémy’s student Auguste Verneuil refined the process, launching the first commercially viable flame fusion technique. By 1902 he published the method, and by 1913 his process was producing 10 million carats (≈2 000 kg) of rubies annually.
The 1918 Czochralski method—crystal pulling—introduced a fast, inexpensive way to grow flawless rubies, especially for laser applications. During World War II, the Linde Division of Union Carbine Corporation extended Verneuil’s process to produce 30‑inch (750 mm) ruby rods for bearings.
Bell Telephone Company’s 1958 hydrothermal method, later refined into the hydrothermal growth technique, employs high temperatures and pressures to grow rubies from seeds in a water‑based solution. The flux method, first commercialized in 1959 by Carroll Chatham, creates a molten magma‑like environment that yields natural‑looking gems over nearly a year.
Methods of Synthesizing
Modern ruby synthesis falls into two primary categories: melt‑based and solution‑based. The most common melt techniques are Verneuil’s flame fusion and the Czochralski crystal‑pulling process. Popular solution methods include flux growth and hydrothermal growth.
- Flame fusion rubies are cost‑effective and widely used for bearings and everyday jewelry such as class rings.
- Crystal‑pulled rubies, priced over $5 per carat, are preferred for laser and maser components.
- Flux rubies, costing $50+ per carat, are suited for fine jewelry.
- Hydrothermal rubies, ideal for strain‑free crystals or large, non‑rod shapes, cater to specialized industrial needs.
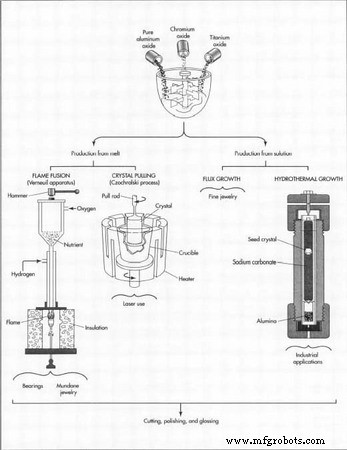 There are several processes used to create synthetic rubies. Verneuil’s flame fusion and Czochralski’s crystal pulling are the most commonly used melt techniques, while flux growth and hydrothermal growth are the most popular versions of solution processes.
There are several processes used to create synthetic rubies. Verneuil’s flame fusion and Czochralski’s crystal pulling are the most commonly used melt techniques, while flux growth and hydrothermal growth are the most popular versions of solution processes.
Raw Materials
The core nutrient is ultra‑pure aluminum oxide (Al₂O₃). Chromium oxide (Cr₂O₃) is added at 5‑8 % to impart the essential red color. For star rubies, a trace 0.1‑0.5 % of titanium dioxide (TiO₂) is incorporated.
Additional chemicals depend on the synthesis method:
- Flame fusion uses an oxygen‑hydrogen torch to melt the powders.
- Czochralski employs electric heating.
- Flux growth dissolves the nutrient in a solvent such as lithium oxide, molybdenum oxide, or lead fluoride.
- Hydrothermal uses an aqueous sodium carbonate solution.
Corrosion‑resistant liners—often silver or platinum—are used in the crucibles and vessels for Czochralski, flux, and hydrothermal processes.
The Manufacturing Process
Crystal Growth
Four primary methods are used to grow synthetic rubies:
- Flame Fusion (Verneuil): Powdered nutrient is fed through a rotating hammer into a high‑temperature flame (≈3 600 °F). The molten material drops onto a ceramic pedestal, where the crystal grows. A typical 12‑15‑carat ruby (≈2.5 g) forms in roughly five and a half hours.
- Czochralski Crystal Pulling: A seed crystal is lowered into molten nutrient within an electric‑heated crucible. The seed rotates and is slowly lifted, pulling a crystal that can reach diameters over 2 in (50 mm) and lengths exceeding 1 m.
- Flux Growth: The nutrient dissolves in a molten flux at 1 470 °F–2 200 °F. Crystals grow over 3–12 months, after which the flux is removed or dissolved.
- Hydrothermal Growth: Nutrient powder sits at the bottom of a pressure‑rated tube, while a seed crystal sits at the top. A heated water‑based solution drives nutrient migration toward the seed under 83,000–380,000 kPa pressure. Growth rates are ~0.15 mm per day.
Surface Finishing
- Polishing: Diamond powder abrasives smooth the surface, leaving only microscopic scratches.
- Glossing: A rapid flame heats the surface to melt micro‑projections, which solidify into a glass‑smooth finish. This treatment can nearly double a ruby rod’s tensile strength.
Comparing Synthetic to Natural
Industrial synthetic rubies are easily identified by their rod shape. Gem‑cut synthetic rubies require microscopic inspection to detect characteristic inclusions, bubbles, and growth bands that distinguish them from natural stones. Advanced analyses can even trace a synthetic ruby back to its manufacturing method.
Manufacturing process
- Mastering Synthetic Monitoring: Proven Best Practices for Reliable SaaS & Web Performance
- Synthetic Rubies: Production Methods, History, and Industrial Applications
- Synthetic PAO Gear Oil Extends Food-Plant Worm Gearbox Life by 350%
- Why 3D‑Printed Synthetic Body Parts Are Revolutionizing Medical Device Development
- Python vs Ruby: A Comprehensive Comparison of Features, Advantages, and Use Cases
- Advances in Synthesis and Applications of Silver Nanostructures
- Comparative Analysis of Electrochemical, Biomedical, and Thermal Properties of Natural vs. Synthetic Nanomaterials
- Polyaniline Nanoskein: Innovative Synthesis, Comprehensive Characterization, and Advanced Redox Sensing
- Industrial Machinery Lubricants: 3 Essential Types Explained
- Synthetic Resin Explained: Properties, Uses, and Benefits



