Hypodermic Syringe: Design, History, and Manufacturing Excellence
The hypodermic syringe, also known as a hypodermic needle, is a precision medical device used by clinicians to transfer liquids into or out of the body. A hollow needle attaches to a tube and a plunger; pulling the plunger draws fluid into the tube, while pushing it out forces the fluid through the needle. Introduced in the mid‑1800s, the syringe has evolved steadily with new materials and designs, becoming an indispensable tool in modern medicine and a symbol of clinical care.
History
Early pharmaceutical advances sparked the quest for efficient drug delivery. In 1809, Francois Magendie demonstrated that drugs could be introduced through the skin using a coated wooden barb. A. J. Lesieur followed in 1825, applying substances directly to skin blisters. G. V. Lafargue furthered this work by injecting morphine under the skin with a lancet, and F. Rynd invented a drip needle in 1844, though he published his method only in 1861.
The first true hypodermic syringe was developed by Alexander Wood in 1853. He modified a birthmark‑treating syringe by adding a needle and using it to deliver morphine to patients with sleep disorders. Adding a graduated scale and a finer needle a few years later made the device more accurate and attractive to the medical community, leading to widespread adoption.
Over time, design innovations such as a glass piston reduced leaks and infection risk, while the advent of plastics in the late nineteenth century lowered costs and enhanced safety. Mass production techniques emerged, further standardizing the syringe’s reliability.
Background
The syringe operates on simple physics: a vacuum created by the plunger draws fluid through the needle into the barrel. The user reads the measurement marks on the barrel to determine the exact volume. When the plunger is pressed, the fluid is expelled through the needle into the subcutaneous tissue, where it is rapidly absorbed into the bloodstream.
The term “hypodermic” derives from the Greek hypo (“under”) and derma (“skin”), accurately describing the device’s function.
Drug delivery via hypodermic syringe offers clear advantages over oral or transepidermal routes: it bypasses digestive degradation, ensures rapid absorption, and reduces the likelihood of immune rejection.
Design
All hypodermic syringes share core components: a barrel, plunger, needle, and cap. The barrel’s diameter and length vary to suit specific applications, while the tapered tip ensures accurate dosage delivery. The plunger features a handle and a rubber head that seals tightly against the barrel, creating an airtight environment that prevents contamination and preserves dose accuracy. The needle’s gauge, length, and attachment method (fixed or interchangeable) depend on the intended use, and a protective cap safeguards against accidental stick injuries.
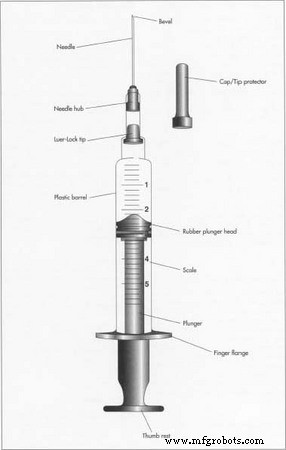 Diagram of a hypodermic syringe. Retraction of the plunger creates the vacuum to draw up materials, which can then be discharged by pushing on the plunger. Its rubber head makes an airtight seal against the walls of the barrel.
Diagram of a hypodermic syringe. Retraction of the plunger creates the vacuum to draw up materials, which can then be discharged by pushing on the plunger. Its rubber head makes an airtight seal against the walls of the barrel.
Raw Materials
Because syringes contact the body’s interior, they must be made from biocompatible, pharmacologically inert materials that can be sterilized and are non‑toxic. Needles are typically fabricated from heat‑treated stainless or carbon steel, often nickel‑plated to resist corrosion. The barrel may be plastic, glass, or a composite, while the plunger handle and rubber head are commonly produced from flexible synthetic rubber.
The Manufacturing Process
Manufacturing follows a consistent sequence: needle formation, plastic component molding, assembly, packaging, labeling, and shipping.
Making the Needle
- 1. Steel is heated until molten, drawn through a precision die, and rolled into a continuous hollow wire. The wire is cut to the required needle length. Advanced needles may be cast directly from a die, and additional metal components are produced similarly.
Making the Barrel and Plunger
- 2. For simple barrel shapes, extrusion molding is employed: plastic or glass granules are fed into a hopper, heated, and forced through a heated chamber and die to form a continuous tube, which is then cooled and cut.
- 3. Complex parts—such as the barrel ends, plunger, and safety caps—are created by injection molding. Plastic is heated into a liquid state, injected into a mold, cooled, and solidified into the final shape. The rubber plunger head can also be molded this way before attachment to the handle.
Assembly and Packaging
- 4. Once components are ready, assembly occurs on a conveyor: the plunger is inserted, barrel ends are capped, and graduation markings are printed with precision calibration to ensure accurate volume measurements. The needle and safety cap are attached.
- 5. Sterility is paramount; each syringe is individually sealed in airtight plastic, then grouped in boxes, stacked on pallets, and shipped to distributors.
Quality Control
Quality assurance occurs at every stage. Random inspections check size, shape, and fit, using visual assessment and precision tools such as vernier calipers, micrometers, and microscopes. Line inspectors monitor production points for deformities, improper assembly, or packaging errors.
Regulation is governed by the U.S. Food and Drug Administration (FDA), which enforces Good Manufacturing Practices, conducts inspections, and requires comprehensive record‑keeping. Manufacturers also maintain their own stringent product specifications.
The Future
Continued research aims to create syringes that are safer, more durable, reliable, and cost‑effective. Innovations include minimal‑processing metals and plastics to reduce waste, accelerate production, and lower costs, while maintaining the highest safety standards.
Manufacturing process
- What is VMC Machining? An Expert Overview of Vertical Machining Centers
- Expert Guide to Aluminum Laser Marking: Precision, Durability, and Industry Applications
- MIG vs. TIG Welding: Selecting the Right Arc Welding Technique for Your Project
- Comprehensive Guide to Laser Marking: Types, Benefits, and Applications
- Key Considerations for High‑Volume Swiss Machining
- Tungsten Needle: Design, Properties, and Applications
- Hypodermic Syringe: Design, History, and Manufacturing Excellence
- Designing with Markforged Composite Filaments: Minimum Feature Sizes for Optimal Reinforcement
- Revolutionizing Needle Production: Moldworx’s Automated Overmolding & Inspection Cell
- NuTec Deploys Epson Cleanroom SCARA Robots for Automated Syringe Production



