Chalk: From Ancient Cave Art to Modern Classroom Standards
Background
Classroom chalk comes in slender sticks that measure roughly 0.35 inches (9 mm) in diameter and 3.15 inches (80 mm) in length. This simple, low‑cost tool has become the standard for conveying information to large groups of students.
Chalk has been used for drawing since prehistoric times. Archaeologists trace the earliest cave paintings to chalk deposits that were applied to walls by early humans. Throughout history, artists across cultures have favored chalk for its ease of use, and many surviving sketches are protected with shellac or similar varnishes.
In the nineteenth century, the rise of larger classrooms demanded a more efficient teaching aid. Chalkboards (originally called blackboards) and individual student chalkboards were introduced, each with a supply of chalk sticks and a sponge or cloth eraser. The transition from ink‑written notes to chalk‑written outlines made lesson delivery faster and more visually engaging.
As chalkboards evolved from slate to synthetic materials, the color palette shifted. While early blackboards were literally black, the 20th‑century shift to green boards was driven by eye‑comfort considerations. Simultaneously, the preferred writing medium became yellow chalk, offering high contrast against the darker board.
Today, nearly all chalk is manufactured as a “dustless” product. While some fine dust still escapes during use, manufacturers harden the chalk through extended baking or coat it with shellac, reducing airborne particles and improving user experience.
Raw Materials
The core ingredient is calcium carbonate (CaCO3), derived from limestone formed by the accumulation of coccoliths—tiny calcareous plates produced by plankton. These deposits build sedimentary layers rich in calcium, which are harvested for chalk production.
For pastel chalks, calcium sulfate (CaSO4)—obtained from gypsum—serves as the base. Gypsum, an evaporite mineral, shares a similar geological origin with limestone. Pastel chalks also incorporate clays, oils, and vibrant pigments, yielding sticks that write smoothly on paper and remain vivid after drying.
The Manufacturing Process
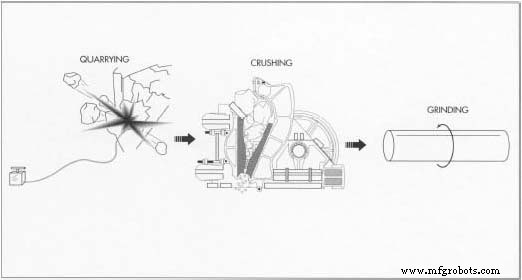
Quarrying Limestone
- Approximately 95 % of U.S. limestone is extracted via open‑pit quarries. After securing a reserve of at least 25 years’ worth, heavy machinery removes surface deposits, and explosives fracture the rock to create workable blocks.
Pulverizing the Chalk
- Large limestone chunks are transported to crushing stations where primary crushers break them into smaller pieces, followed by secondary crushers that produce pebbles.
- Wet grinding in ball mills further refines the material, washing away impurities and producing a fine powder suitable for chalk.
- After grinding, the powder is sieved on vibrating screens to isolate the finest particles. The material is mixed with water, extruded through a die, cut to length, and cured in an oven for four days at 188 °F (85 °C).
Dehydrating Gypsum
- Gypsum is quarried, crushed, and then heated in a kettle to 244–253 °F (116–121 °C) until its moisture content drops by 12–15 %. A subsequent heating step to 402 °F (204 °C) removes the remaining water, leaving calcium sulfate.
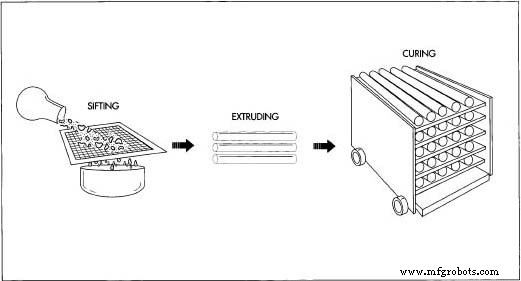
Sifting, Cleaning, and Shipping
- Both chalk and calcium sulfate powders are further refined on vibrating screens, washed, dried, bagged, and shipped to manufacturers. Upon arrival, the material is re‑ground to ensure uniform smoothness.
Making White Classroom Chalk
- A thick slurry of calcium carbonate and water is extruded into long, thin sticks. The sticks, originally 24.43 inches (62 cm) long, are cut to 80 mm, placed on a sheet, and baked for four days at 188 °F (85 °C).
Making Colored Classroom Chalk
- Pigments are mixed with dry calcium carbonate before water is added. The resulting slurry undergoes the same baking process as white chalk, producing a range of vibrant colors.
Making Pastels
- Pastels use calcium sulfate as the base, combined with clays, oils, and a higher pigment load. The mixture forms a toothpaste‑like consistency and is air‑dried rather than baked to preserve moisture.
Boxing the Chalk
- Completed sticks are stacked in small boxes and then shipped in larger cartons to retailers.
Quality Control
Classroom chalk must meet stringent safety and performance standards. All raw materials undergo purity tests before use. After shaping, a representative stick from each batch is evaluated for density, break strength, marking quality, and erasability. The chalk is also tested for dust retention after cleaning the board.
Chalk that satisfies the American National Standards Institute (ANSI) criteria receives the “CP” (Certified Product) nontoxic seal from the Art & Craft Materials Institute. This certification confirms compliance with voluntary safety standards and ensures that the product’s ingredients, labeling, and manufacturing processes are transparent and safe for children and adults alike.
The Future
While digital whiteboards and interactive displays have gained popularity, many educators still favor traditional chalkboards for their tactile immediacy and low cost. Studies comparing overhead projectors with chalkboards have found that the latter foster greater student interaction and engagement.
Electronic chalkboards—large screens that replicate the look of a chalkboard—offer visual appeal and multimedia capabilities. However, high upfront costs, limited interactivity, and teacher preparation time often outweigh the benefits for many schools. Consequently, the humble chalk is likely to remain a staple in classrooms for the foreseeable future.
Manufacturing process
- Titanium: Key Properties, Applications, and Industrial Uses
- Castanets: History, Craftsmanship, and Performance
- The History, Production, and Future of Glue: From Ancient Adhesives to Modern Innovations
- Thread: Types, History, Production, and Quality Control
- Acetylene: Production, Uses, and Handling – A Comprehensive Overview
- Comprehensive Overview of Asbestos: From Ancient Uses to Modern Health Risks
- The History, Design, and Manufacturing of Dice: From Ancient Tools to Modern Gaming
- Plywood: History, Production, and the Path Ahead
- Tin: From Bronze Age to Modern Industry – Uses, Production, and Future
- Silver: History, Properties, and Modern Applications



