Aluminum Alloys: Protecting Ship Hulls From Corrosion

Aluminum Alloys: Protecting Ship Hulls From Corrosion
Effective corrosion protection of an aluminum‑alloy hull directly determines a vessel’s service life. While the material’s intrinsic resistance is vital, the chosen repair and coating strategies ultimately decide how well the hull survives the harsh marine environment.
Cladding Protective Layer
In the mid‑1960s, Chinese shipbuilders tested oxy‑acetylene flame‑sprayed polychloro‑ether plastics and vinyl chloride paint on aluminum‑alloy hulls. Both coatings failed: the plastics peeled off the anodized surface, and the paint could not adhere to the hull’s bottom, flaking within weeks of sea exposure. Consequently, these films were deemed unsuitable for long‑term protection.
Later, GNA neoprene (chloroprene) rubber proved highly effective. Its chemical compatibility with aluminum creates a robust barrier, allowing continuous protection for over a decade without significant maintenance—an impressive reduction in upkeep costs for commercial operators.
Cladding protective layer with GNA neoprene rubber
Paint Coating
Paint remains the most widespread defense for aluminum hulls and superstructures. The performance of anti‑corrosion paint depends heavily on raw‑material quality and application technique. Prior to full application, a small‑sample cavitation test ensures surface readiness; only qualified surfaces receive paint.
Epoxy‑zinc yellow coatings are simple to apply, but phosphating primers require meticulous surface cleaning, controlled temperature, and humidity. When executed correctly, these coatings provide a durable barrier against chloride attack.
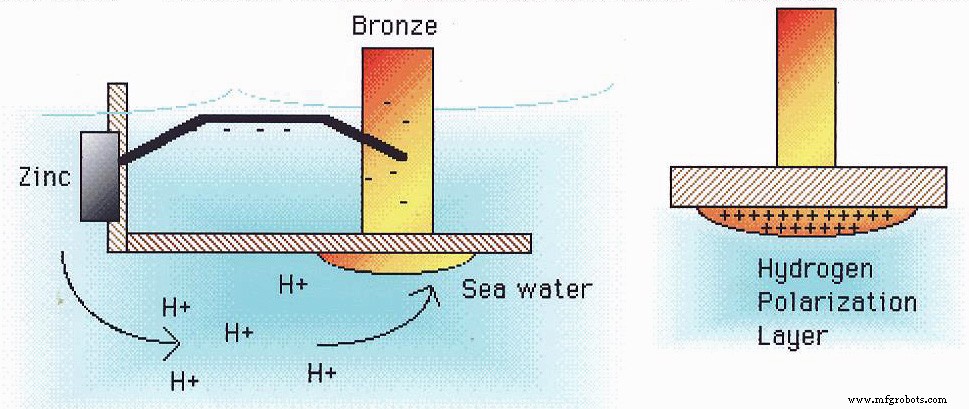
Anodic Sacrifice Protection
Seawater’s electrolyte properties create an “electrolytic tank” around the ship. Copper alloy thrusters, steel shafts, and rudders exhibit more positive potentials than aluminum, which acts as an anode. Simply painting the hull does not stop the galvanic corrosion inherent in this configuration.
Today, the most effective strategy combines sacrificial anode systems with paint coatings. Low‑potential zinc or zinc‑alloy strips, and occasionally magnesium, are installed below the waterline. These anodes corrode preferentially, shielding the aluminum hull (cathode) from oxidation. Together, they extend hull life and reduce repair frequency.
For further information on aluminum alloy performance and advanced corrosion protection, visit SAMaterials.
Metal
- Titanium‑Aluminum Alloys: Advanced Preparation Techniques and Key Industrial Applications
- Aluminum Alloy vs. Copper Cables: Which Is the Superior Choice?
- Protecting Aluminum Against Corrosion: Expert Strategies for Long‑Lasting Durability
- Carbon Steel vs Alloy Steel: Key Differences Explained
- Securing Your IoT Product: A Practical Guide to Preventing Hacker Attacks
- Aluminum vs. Steel in Fabrication: Performance, Cost, and Application Guide
- Does Aluminum Rust? Key Differences, Types, and Protection Tips
- Preventing Metal Corrosion: Proven Strategies for Long-Lasting Durability
- Preventing Aluminum Corrosion: Proven Strategies for Long-Lasting Durability
- Secure Your Heavy Equipment: Proven Strategies to Prevent Theft



