Vitamins: Essential Nutrients, History, Manufacturing, and Future
Background
Vitamins are essential organic compounds that the body requires in trace amounts to maintain health and physiological function. Deficiencies lead to a spectrum of diseases—from night blindness due to vitamin A deficiency to scurvy from lack of vitamin C and rickets caused by inadequate vitamin D. To date, thirteen vitamins are universally recognized as indispensable, with additional vitamin‑like compounds under investigation.
Historically classified alphabetically—A, B, C, D, E, K—vitamins are chemically diverse. The B‑vitamin cluster, once considered a single entity, has been subdivided into eight distinct nutrients: thiamine (B1), riboflavin (B2), niacin (B3), pantothenic acid (B5), pyridoxine (B6), biotin (B7), folic acid (B9), and cobalamin (B12). These are sourced from plant and animal foods, and can also be produced synthetically; the chemical structure is identical regardless of origin.
While the clinical consequences of deficiency are well established, the potential therapeutic roles of high‑dose supplementation remain a topic of debate. Current consensus emphasizes adequate intake per recommended daily allowances rather than supraphysiologic dosing.
History
The concept of vitamins emerged in the early 20th century. English physician William Prout (1827) first delineated the three macronutrient groups—fats, carbohydrates, and proteins. In 1906, Frederick Hopkins demonstrated that mice fed a diet of these essentials alone failed to thrive without additional trace nutrients. Polish scientist Casimir Funk coined the term "vitamines" in 1912, deriving it from the Latin "vita" (life) and the Greek "amine," reflecting early assumptions that vitamins were amines.
Subsequent isolation of individual vitamins—such as thiamine, riboflavin, and vitamin C—confirmed that these compounds were chemically distinct. Over the decades, the full spectrum of thirteen essential vitamins was identified and synthesized, laying the groundwork for modern nutrition science.
Raw Materials
Vitamins can be extracted from natural sources or synthesized in the laboratory. For example, vitamin A is commonly derived from fish liver oil, while vitamin C is extracted from citrus fruits or rose hips. Synthetic production offers cost‑effectiveness and scalability; for instance, vitamin A can be synthesized from acetone, and vitamin C from keto‑acid precursors. Chemically, there is no difference between natural and synthetic forms.
Supplement tablets and capsules typically contain excipients that facilitate manufacturing and absorption. Common fillers include microcrystalline cellulose, lactose, calcium, and maltodextrin. Lubricants such as magnesium stearate or stearic acid ensure smooth tablet flow, while silicon dioxide acts as a flow agent. Disintegrants like modified cellulose gum or starch promote rapid dissolution after ingestion. Coatings—often cellulose‑based and sometimes enhanced with carnauba wax—improve palatability, protect against gastric acid (enteric coating), or control release kinetics.
Herbal additives and minerals (e.g., calcium, iron, zinc) are sometimes incorporated. The supply chain typically involves specialized laboratories purifying vitamins and minerals, distributors supplying manufacturers, and final product assemblers producing multivitamin tablets or B‑complex capsules.
The Manufacturing Process
Preliminary Check
- Raw materials are sourced from vetted distributors and accompanied by a Certificate of Analysis confirming identity and potency. Manufacturers may conduct independent laboratory testing, particularly for herbal ingredients, to verify purity and detect bacterial contamination.
Preblending
- Most raw vitamins arrive as fine powders requiring minimal processing. If coarser, they are milled. Preblending with a filler (e.g., microcrystalline cellulose) can improve granule uniformity before downstream steps.
Wet Granulation
- Wet granulation improves particle size distribution, essential for tabletability. The powder is mixed with cellulose particles and wetted, then dried and milled into uniform granules.
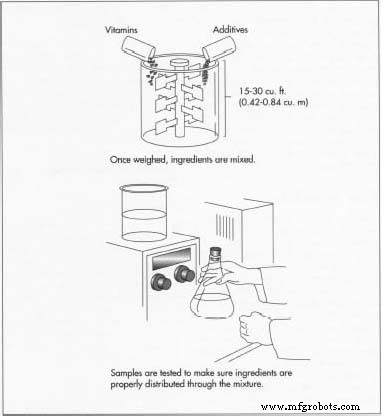
Weighing and Mixing
- Ingredients are weighed according to the batch formula, then blended in a 15–30 cu ft (0.42–0.84 cu m) mixer for 15–30 minutes. During blending, samples are tested to ensure homogeneous distribution. The finished blend is then transferred to the encapsulating or tableting line.
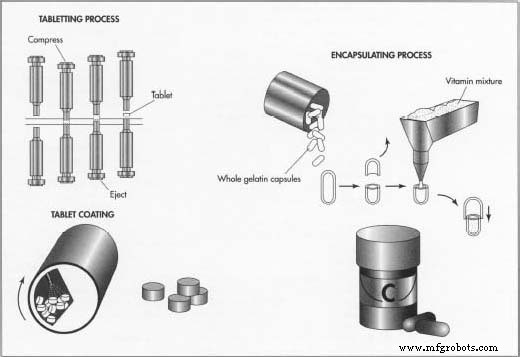
Encapsulating Machine
- The blend is fed into the encapsulator, where gelatin capsules are opened, filled with the precise powder quantity, and closed. Visual and weight checks ensure capsule integrity.
Polishing and Inspection
- Filled capsules undergo polishing to remove excess dust, then pass through an inspection table that rotates the capsules for full visual assessment. Imperfect capsules are removed before packaging.
Tableting
- Tablet production occurs on a tableting machine where the powder is transferred from a hopper to a rotating table with interchangeable dies. The powder is compressed under 4–10 tons of pressure to form tablets of desired shape. The speed of rotation dictates production rate, and a vibrating belt removes dust before the tablets move to the coating area.
Coating
- Tablets are coated in a rotating pan with spray guns, often receiving a secondary carnauba wax layer. Coatings can mask taste, provide color, protect against gastric acid (enteric), or control release. After drying, tablets are ready for packaging.
Packaging
- Automated packing lines handle capsule or tablet counting, bottling, capping, labeling, shrink‑wrapping, and boxing. Human intervention is limited to setting machine parameters.
Quality Control
Quality assurance occurs at every stage: raw material testing, blend uniformity, post‑tablet inspection, and final product analysis. Federal regulations dictate permissible ingredients and allowable health claims. All ingredients must be proven safe before consumer release.
The Future
Ongoing research continually refines our understanding of vitamin functions and therapeutic potential. While public interest may surge around newly implicated nutrients, the core manufacturing processes remain stable. Advances are likely to focus on precision nutrition, personalized dosing, and deeper mechanistic insights rather than radical changes in production.
Manufacturing process
- What is VMC Machining? An Expert Overview of Vertical Machining Centers
- Expert Guide to Aluminum Laser Marking: Precision, Durability, and Industry Applications
- MIG vs. TIG Welding: Selecting the Right Arc Welding Technique for Your Project
- Comprehensive Guide to Laser Marking: Types, Benefits, and Applications
- Key Considerations for High‑Volume Swiss Machining
- CNC Prototyping Guide: Precision, Speed, and Cost‑Effective Production
- Expert Shaft Manufacturing: Precision Processes & Custom Solutions
- Fiber Laser Marking: Precision, Speed, and Versatility for Modern Industries
- Zirconium Metal: The Industrial Powerhouse
- Vitamins: Essential Nutrients, History, Manufacturing, and Future



