Optimizing Pd/SnO₂ Nanomaterial Synthesis for High‑Performance Hydrogen Gas Sensors
Abstract
Metal‑oxide hydrogen sensors were fabricated using nanosized tin dioxide prepared by a sol‑gel route. The sensing layers were impregnated with PdCl₂ solutions of varying concentrations to enhance sensitivity. We investigated how the temperature profile during sensor sintering influences the nanomaterial’s particle size, which in turn dictates sensor performance. Shorter high‑temperature dwell times prevent particle coarsening, yielding smaller grains that deliver higher responses to 40 ppm H₂. The data highlight the critical role of the Pd/SnO₂ interfacial boundary length in governing gas‑sensing behavior. The resulting sensors exhibit rapid response/recovery and stable long‑term operation.
Background
Hydrogen is a key feedstock in chemical synthesis and a clean energy carrier, yet its flammability demands stringent monitoring during production, transport, and storage [1–3]. Metal‑oxide sensors offer a promising platform for real‑time H₂ detection [4–6].
Nanostructured SnO₂ possesses desirable attributes—chemical inertness, thermal stability, and robust O₂ chemisorption—making it an attractive material for hydrogen sensing [7–9]. Sensitivity can be further amplified by incorporating catalytic Pd, a well‑known facilitator of H₂ oxidation [6,10].
The physicochemical properties of the sensing layer depend heavily on the synthesis route, composition, and sintering conditions, which govern particle size and morphology—critical factors for sensor efficacy [11–18].
While high‑temperature sintering is required for mechanical integrity and electrical conductivity, it tends to promote particle growth. Optimizing sintering schedules can suppress coarsening while maintaining robust nanoparticle contacts [18].
Our study focuses on how varying Pd loading and sintering protocols affect Pd/SnO₂ nanomaterial characteristics and, consequently, hydrogen sensor performance.
Methods
Synthesis of Nanosized Tin Dioxide
SnCl₄·5H₂O (1.5 g) was dissolved in 15 mL ethylene glycol, evaporated at 110–120 °C to yield a dark brown gel, then dried at 150 °C to form a xerogel. Thermal decomposition in air produced ~8 nm SnO₂ particles. The resulting nanomaterial, carboxymethyl cellulose, and PdCl₂ were blended to prepare the sensing layer.
Preparation of Adsorption‑Semiconductor Sensors
A 3 μL paste (SnO₂ + 3 wt% CMC) was deposited on a ceramic substrate with integrated contacts and heater [19]. The paste yielded a ~70 μm thick layer (SEM data). Sensors were dried at 90 °C for 1 h. Palladium impregnation was performed with PdCl₂ solutions (0.05 × 10⁻²–0.15 M). Post‑impregnation, sensors were sintered using two distinct temperature profiles (Fig. 1). Sensors sintered via Mode 1 were labeled S₁; those via Mode 2 were labeled S₂.
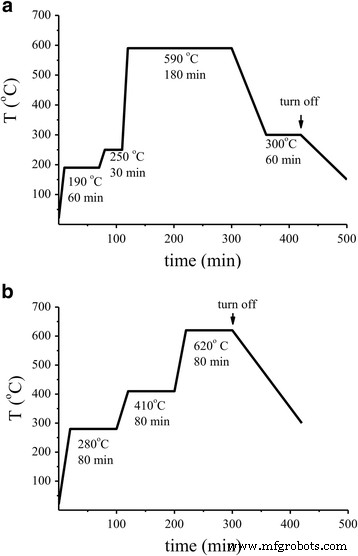
Schemes of temperature heating of the sensors based on SnO₂. a Mode 1. b Mode 2
Methods of Measurement
Sensors were tested in 400 mL/min gas streams within a dedicated chamber. Heater voltage controlled operating temperature, verified by an Optris Laser Sight pyrometer. Prior to measurement, sensors were aged at 400 °C for 1 week, with periodic exposure to 1000 ppm H₂.
Sensor response was defined as R₀/R_H₂, where R₀ is resistance in air and R_H₂ is resistance in 40 ppm H₂. Response time (t₀.₉) and recovery time (τ₀.₁) were measured by the time to reach 90 % of the equilibrium signal upon gas exchange.
Gas mixtures containing H₂, CO, CH₄, and their combinations were prepared at the Ukrainian Centre of Certification and Metrology to assess selectivity.
Long‑term stability was evaluated by measuring the 40 ppm H₂ response over 6 months for sensors S₂ (S‑67, S‑69).
Specific surface area was measured by BET. Palladium content was quantified via atomic absorption (AAS1N Carl Zeiss) using an acetylene‑air flame. Phase composition was characterized by XRD (Bruker D & Advance, Cu Kα). TEM (SELMI PEM‑125 K, 100 kV) and FESEM (JEOL JSM‑6700F) provided morphological details; HRTEM (JEM‑2100F) resolved interparticle boundaries. Layer thickness was determined by SEM (JEOL JSM‑6060LA).
Results and Discussion
The synthesized ~8 nm SnO₂ nanomaterial served as the base for all sensors. Comparison of sintering protocols revealed that Mode 1 (590–620 °C, 180 min) produced particles averaging 17 nm (Fig. 2a) with a sensor response of 6.7. Shortening the dwell time to 80 min at 590 °C drastically increased resistance (>500 MΩ) and reduced response (~2), indicating insufficient interparticle contact.
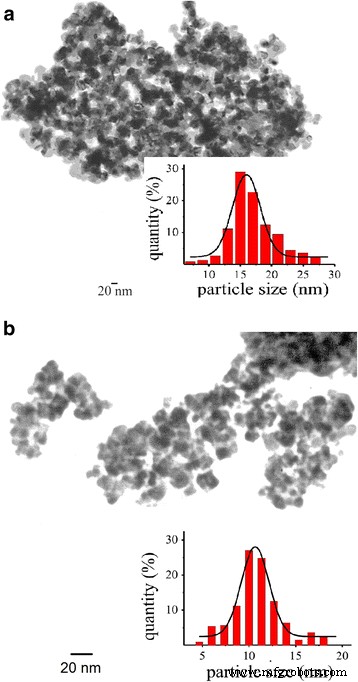
TEM images of the gas‑sensitive nanomaterials a S₁ and b S₂
Mode 2 introduced a prolonged low‑temperature phase (280 °C and 410 °C) to foster particle contact formation, followed by a 620 °C step. This yielded smaller grains (average 11 nm) and a higher BET surface area (47 m²/g vs. 39 m²/g for S₁). Pd loading increased linearly with the precursor concentration (0.001–0.193 wt % for 0.05–0.15 M PdCl₂).
XRD confirmed a cassiterite structure (a = 0.4738 nm, b = c = 0.3187 nm) across all samples. FESEM and HRTEM images (Fig. 3) revealed well‑defined SnO₂ grains and dispersed Pd nanoparticles, with clear interparticle boundaries.

a FESEM and b, c HRTEM images of the sensor Pd/SnO₂ nanomaterial
Electrical resistance in air displayed a complex dependence on Pd content: a low‑Pd minimum and a high‑Pd maximum were observed for both sintering modes (Fig. 4). The initial resistance drop at ≤0.05 % Pd likely reflects metallic Pd’s conductivity. At higher Pd loadings, reduced oxygen chemisorption at the Pd–SnO₂ interface leads to increased resistance; beyond a threshold, Pd aggregation shortens interfacial boundaries, lowering resistance again.
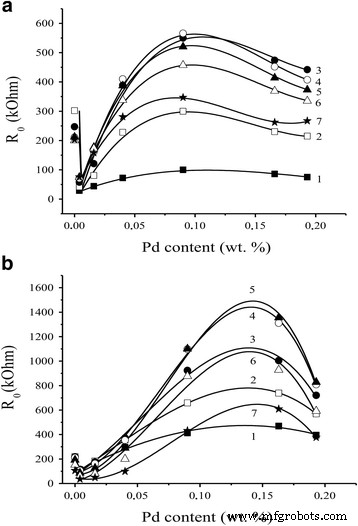
Dependence of R₀ values of sensors a S₁ and b S₂ on Pd content at various temperatures (410–225 °C).
Higher resistance in air correlates with enhanced H₂ response (Fig. 5). Sensors S₂ outperformed S₁, achieving a response of 19.5 at 40 ppm H₂ and 261 °C. Excessive Pd (≥0.15 %) diminishes response due to cluster aggregation covering SnO₂ surface sites.
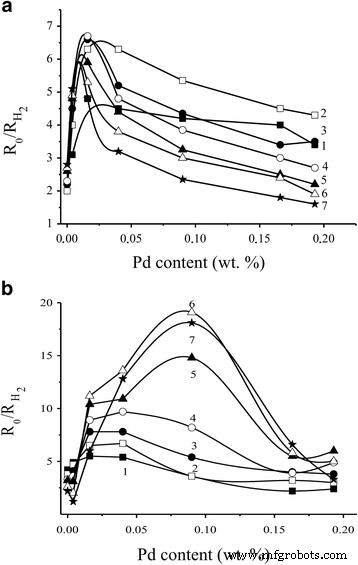
Dependence of the sensor response to 40 ppm H₂ for sensors a S₁ and b S₂ on Pd content at various temperatures.
The optimally doped sensor (0.09 % Pd/SnO₂) exhibited linear conductivity over 2–1000 ppm H₂ at 327 °C and 382 °C, with a detection limit of 2 ppm (R₀/R_H₂ = 2.1 at 261 °C). Response and recovery times were 3 s and 12 s, respectively—remarkably fast compared to prior reports (120 s/15 min) [31].
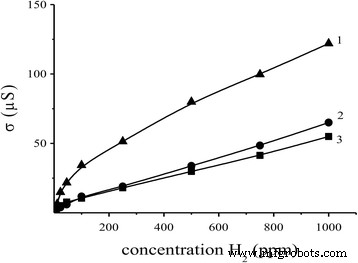
Conductivity of the sensor S₂ (0.09 % Pd/SnO₂) versus H₂ concentration at 261, 327, and 382 °C.
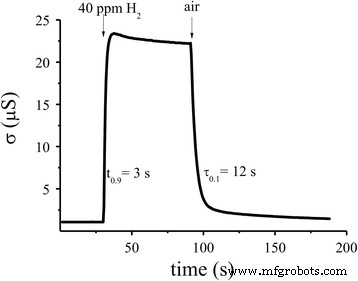
Dynamic response of the optimal Pd‑doped sensor (0.09 % Pd/SnO₂) at 261 °C.
Selectivity tests (500 ppm CO/CH₄) demonstrated a markedly higher response to H₂ compared to CO and CH₄, confirming minimal cross‑interference at 261 °C. The lower optimal temperature for H₂ relative to CH₄ (382 °C) and CO (327 °C) reflects the superior catalytic activity of H₂ on Pd/SnO₂ surfaces.
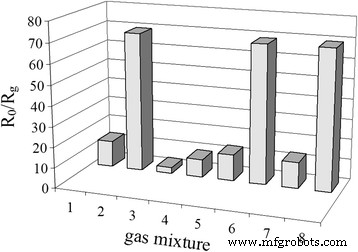
Response of sensor S₂ (0.09 % Pd) to various gas mixtures at 261 °C.
Long‑term stability over 6 months showed no drift or loss of sensitivity for S₂ (S‑67, S‑69) (Fig. 9), underscoring the robustness of the sintering strategy.
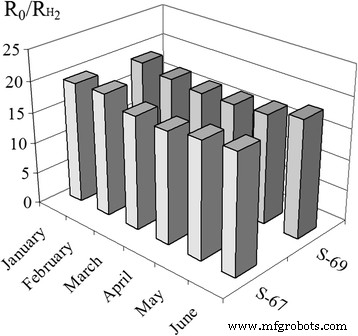
Stability of the 40 ppm H₂ response for sensors S₂ (S‑67 and S‑69) over 6 months at 261 °C.
Conclusions
Tailoring the high‑temperature sintering regime for Pd/SnO₂ sensors yields sub‑10 nm grains that dramatically enhance hydrogen response (R₀/R_H₂ = 19.5) at 261 °C. These sensors detect H₂ from 2 to 1089 ppm with a 2 ppm detection limit, exhibit 3 s response and 12 s recovery, and maintain stability over extended use.
Nanomaterials
- Key Automotive Sensors: How Modern Cars Use Advanced Technology
- Digital Magnetic Hall Sensors: Fundamentals, Design, and Automotive Applications
- UV-Enhanced Copper Oxide Nanowire Sensor for Rapid Hydrogen Detection at Room Temperature
- Perpendicular Magnetic Anisotropy and Hydrogen‑Induced Magnetization Modulation in Ta/Pd/CoFeMnSi/MgO/Pd Multilayers
- Ultra‑Sensitive Room‑Temperature CNTs/SnO₂/CuO Sensor for Rapid H₂S Detection
- Deep Dive into MEMS Actuation & Sensing Mechanisms: Your Guide to Advanced Sensor Design
- Digital Sensors in Industrial Machinery: Function, Types, and Best Practices
- NASA-Developed Inductive Non-Contact Position Sensor for Precise Motion Control
- New Sensors Safeguard Hydrogen Vehicles by Eliminating Sparking
- Distance Sensors Explained: Types, Uses, and How to Choose the Right One



