Tungsten Nanolayer Coating Enhances Silicon Anode Performance in Lithium‑Ion Batteries
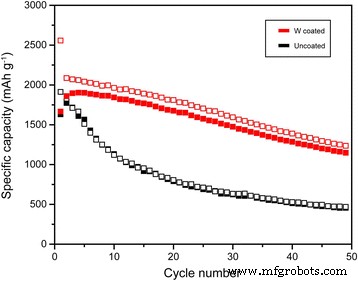
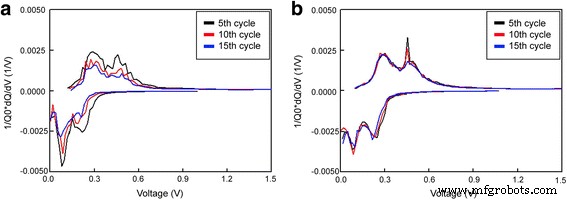
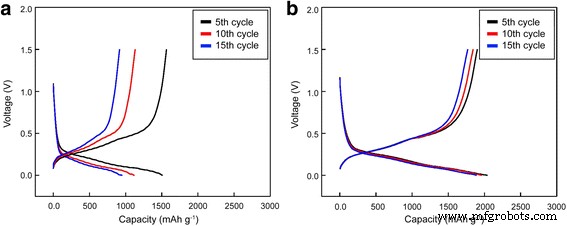
In this work, a 40‑nm tungsten (W) nanolayer was applied to a silicon (Si) anode via physical vapor deposition (PVD). Scanning electron microscopy (SEM), transmission electron microscopy (TEM), energy‑dispersive X‑ray spectroscopy (EDX), and electron probe X‑ray microanalysis (EPMA) confirmed a uniform, dense W coating that enveloped the Si nanopowder (≤ 100 nm). Electrochemical tests in CR2032 coin cells revealed first‑charge capacities of 2 588 mAh g⁻¹ for the W‑coated cell versus 1 912 mAh g⁻¹ for the uncoated reference. After 50 cycles, the W‑coated electrode retained 61.1 % of its initial capacity, compared with only 25.5 % for bare Si. The enhanced cyclability is attributed to the high conductivity and mechanical resilience of the atomic‑scale W layer, which buffers volume expansion and preserves electrical contact. Silicon’s theoretical specific capacity (4 200 mAh g⁻¹) is roughly ten times that of graphite, positioning it as a highly attractive anode material for next‑generation lithium‑ion batteries. However, Si undergoes a 300 % lattice expansion during lithiation, leading to cracking, loss of active material, and deteriorated electrical connectivity. Its intrinsic low electrical conductivity further limits practical use. Consequently, extensive research has focused on mitigating these challenges through composite architectures, nanostructuring, alloying, and surface coatings. Conductive coatings—including carbon, metal alloys, and conductive polymers—have been shown to buffer mechanical strain and enhance electron transport, yet many such approaches involve complex fabrication steps that impede commercial scalability. Physical vapor deposition (PVD) offers a versatile, scalable route to deposit uniform thin films of virtually any inorganic material at the nanometer scale. Compared with chemical deposition, PVD generates lower interfacial resistance and produces films with superior hardness and wear resistance. Tungsten, in its pure form, possesses the highest tensile strength and hardness among metals, making it an ideal candidate for a protective, conductive layer on Si anodes. In the present study, we employed PVD to deposit a tungsten nanolayer on Si electrodes, then evaluated the resulting structural integrity and electrochemical performance using a suite of microscopy and spectroscopy techniques. Silicon electrodes were prepared by mixing 40 wt % Si nanopowder (≤ 100 nm), 40 wt % Denka Black (conductive carbon), and carboxymethyl cellulose binder in deionized water to form a slurry. The slurry was cast onto 50 µm copper foil, then dried at 70 °C for one hour. Tungsten was sputter‑coated onto the Si surface at 100 °C in argon plasma for 5 min using a PVD system (Dongwoo Surface Tech Co., Ltd.). The resulting films were characterized by SEM, TEM, EPMA, and EDX. Schematic of physical vapor deposition for W coating Coin cells (CR2032) were assembled in a dry room. Si anodes were punched to 14 mm diameter; lithium counter electrodes were 16 mm. The W nanolayer weight per 14 mm electrode was ~0.0001 g. The electrolyte was 1 M LiPF₆ in an equal‑volume mixture of ethylene carbonate, dimethyl carbonate, and ethylene methyl carbonate. Cells were rested for 24 h at 40 °C before testing. Galvanostatic cycling was performed on a WBCS 3000 (WonATech Inc.) between 0 and 1.5 V. After cycling, surface morphologies were examined and electrochemical impedance spectroscopy (EIS) was carried out from 10⁻² to 10⁵ Hz with a 5 mV AC amplitude (SOLATRON SI1280B). Figure 2 displays SEM images of pristine (a) and W‑coated (b) Si electrodes. The nanopowder retained its original size, but the W layer increased the particle diameter to ~100–120 nm. EDX mapping (Fig. 2b,d) confirms the presence of W, while EPMA (Fig. 3) shows a uniform distribution. Scanning electron microscopy images and energy dispersive X‑ray profiles of pristine uncoated (a,c) and coated (b,d) Si electrode surfaces TEM cross‑section (Fig. 4) reveals a ~40 nm thick W film that fully encapsulates the Si particles and fills interparticle gaps, ensuring a continuous, mechanically robust coating. Transmission electron microscopy image and depth profiling of the W‑coated Si electrode EIS measurements (Fig. 5) demonstrate that the W coating reduces both the solution resistance (Rₛ) and the charge‑transfer resistance (R_ct) while slightly increasing the surface resistance (R_sei). The lower R_ct values correlate with the higher conductivity of tungsten, which facilitates faster Li⁺ transport during charge. Electrochemical impedance spectroscopy for uncoated and W‑coated Si electrodes before cycling and the corresponding equivalent circuit Cycling performance (Fig. 6) shows a dramatic improvement for the W‑coated cell: first‑cycle charge capacity of 2 588 mAh g⁻¹ versus 1 912 mAh g⁻¹ for bare Si. At 0.1 C, discharge capacities after 10, 20, and 50 cycles are 1 843, 1 676, and 1 137 mAh g⁻¹ (retention 99.1 %, 90.1 %, 61.1 %), compared with 1 132, 790, and 452 mAh g⁻¹ (retention 63.9 %, 44.6 %, 25.5 %) for the uncoated electrode. Charge/discharge capacity profiles for uncoated and W‑coated Si electrodes at 0.1 C over 50 cycles dQ/dV curves (Fig. 7) reveal that the reaction peaks for both electrodes remain in the same voltage windows, indicating that the W layer does not alter the lithiation chemistry. However, the uncoated Si shows increasing polarization and peak shifts with cycling, whereas the W‑coated Si maintains stable reaction potentials, underscoring the buffering effect of the coating. dQ/dV curves for uncoated and W‑coated Si electrodes at 0.1 C over 5, 10, and 15 cycles Voltage profiles (Fig. 8) further confirm that the W‑coated electrode preserves its capacity and reaction voltages over successive cycles, while the uncoated Si exhibits larger voltage drops. Voltage profiles for uncoated and W‑coated Si electrodes at 0.1 C over 5, 10, and 15 cycles SEM after 10 cycles (Fig. 9) shows that the bare Si electrode develops pronounced cracks, whereas the W‑coated Si remains largely intact, confirming the mechanical integrity afforded by the tungsten nanolayer. SEM images of uncoated and W‑coated Si electrodes after 10 cycles Depositing a ~40‑nm tungsten nanolayer onto a silicon anode by PVD markedly improves its electrochemical behavior. The W coating is uniformly distributed, highly conductive, and mechanically robust, enabling the Si electrode to retain 61.1 % of its capacity after 50 cycles—more than double the 25.5 % retention of the uncoated counterpart. The tungsten layer buffers volumetric changes, preserves electrical contact, and reduces charge‑transfer resistance, making this simple, scalable approach a promising pathway for high‑performance Si‑based anodes.Abstract
Background
Experimental
Fabrication of Electrodes
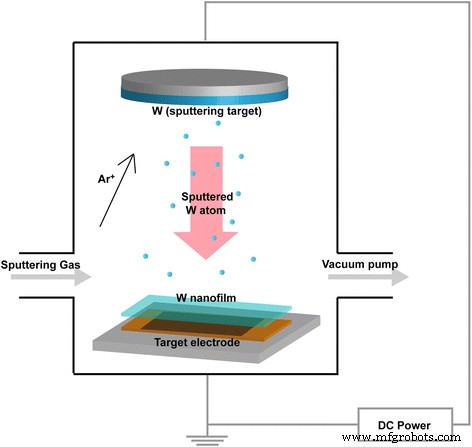
Test Cell Procedure
Results and Discussion
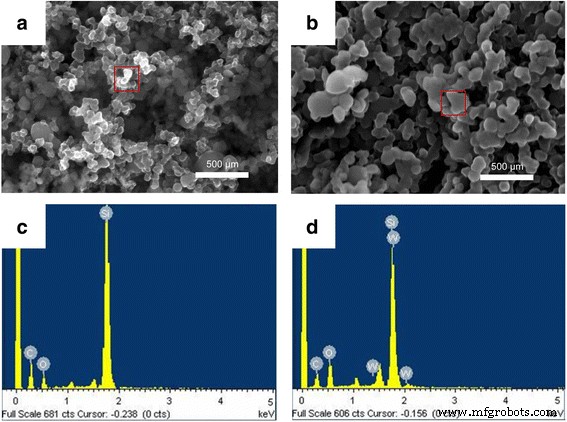
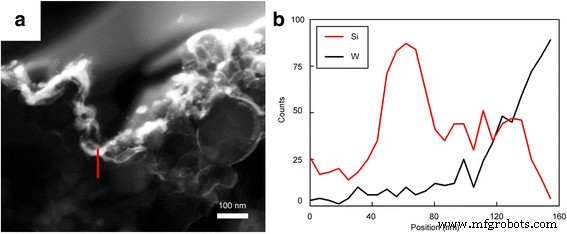
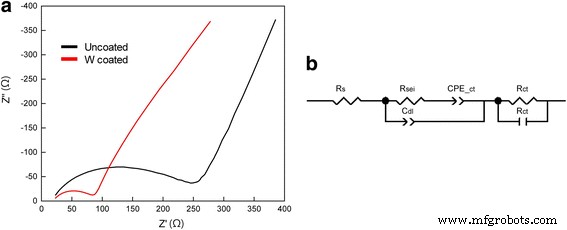

Conclusions
Nanomaterials
- Why Tungsten‑Copper Electrodes Excel in Electrical Discharge Machining (EDM)
- Barium Tungsten Electrodes: Enhancing Xenon Flash Lamp Performance
- Advanced Rare‑Earth‑Doped Tungsten Electrodes for Superior Welding Performance
- Tesla Secures Contract to Build 129‑MWh Lithium‑Ion Battery, the Largest in the World, in South Australia
- Optimizing MAPbI3 Perovskite Solar Cells with Oblique-Angle Sputtered ITO Electrodes
- Binder‑Free 3‑D MoS₂/Graphene Aerogel Anode Delivering 1,041 mAh g⁻¹ for Lithium‑Ion Batteries
- Enhancing Lithium‑Ion Battery Anodes: Deep Cryogenic Activation of Hemp‑Stem Derived Carbon
- Unlocking Growth: Key Opportunities for Lithium‑Ion Battery Manufacturers
- How Lithium‑Ion Batteries Work: A Clear Guide to Their Power
- Boosting EDM Carbide Machining: Superior Wear Resistance with Copper Tungsten Electrodes



