Krypton: Properties, Production, and Uses
Background
Krypton (atomic number 36) is one of the noble gases, sharing the group with helium, neon, argon, xenon, and radon. The gas is colorless, tasteless, and odorless under standard conditions. Its density—about 0.5 oz per gallon (3.7 g L⁻¹)—makes it roughly three times heavier than air. When cooled to extremely low temperatures, krypton liquefies or solidifies; its boiling point is –243.81 °F (–153.23 °C) and its freezing point is –251.27 °F (–157.37 °C).
In nature, krypton occurs as a mixture of six stable isotopes. Isotopes are atoms that share the same number of protons but differ in neutron count, which determines their atomic weight. The most abundant isotope, krypton‑84 (48 neutrons), accounts for 57 % of natural krypton. The other stable isotopes are krypton‑86 (17.3 %), krypton‑82 (11.6 %), krypton‑83 (11.5 %), krypton‑80 (2.25 %), and krypton‑78 (0.35 %).
Krypton can also be produced as a set of unstable, radioactive isotopes during nuclear reactions. Approximately 20 such isotopes have been created in laboratories; all except krypton‑85 have half‑lives of only a few hours. Krypton‑85, with 49 neutrons, is comparatively long‑lived, decaying with a half‑life of 10.73 years.
Industrial applications of krypton are diverse. When blended with argon, it enhances the brightness of fluorescent lamps. In incandescent bulbs, a mixture of krypton and nitrogen extends the filament life. Flashbulbs rely on the rapid release of krypton to generate a brief, intense flash for high‑speed photography. Additionally, krypton‑85’s radioactivity is exploited in nondestructive testing: the gas accumulates in micro‑cracks in metal, allowing flaw detection through its emissions.
History
The noble gases were unknown to early chemists. In 1785, Henry Cavendish detected an inert component in air that did not react with nitrogen. The first definitive identification came in 1868 when Joseph Norman Lockyer, analyzing sunlight, discovered helium and named it after the Greek word for sun.
In 1894, Lord Rayleigh and William Ramsay discovered argon by noting a density difference between nitrogen from the atmosphere and nitrogen from ammonia. They isolated the inert gas, naming it from the Greek word for “inactive.” The following year, Ramsay and Morris Travers observed that heating clevite released helium and argon, marking the first terrestrial detection of helium. In 1898, the duo cooled air to a liquid and separated three new gases—krypton (from Greek “hidden”), neon (“new”), and xenon (“strange”).
In 1900, Friedrich Dom reported that radium decay released helium and a previously unknown radioactive gas. By 1910, Ramsay and Robert Whytlaw‑Gray measured its density and christened it niton (Latin for “to shine”) because it glowed when liquefied; it was later renamed radon. Ramsay received the Nobel Prize in Chemistry in 1904 for his noble gas research.
Initially called rare or inert gases, it was later shown that they are abundant in the atmosphere and can form compounds. In 1962, Neil Bartlett synthesized xenon hexafluoride, the first noble‑gas compound; krypton compounds followed in 1963. The period from 1960 to 1983 saw the meter defined by the wavelength of krypton‑86’s orange‑red emission; the definition was later replaced by the speed of light.
Raw Materials
While trace amounts of krypton appear in various minerals, atmospheric air remains the primary source. At sea level, dry air composition is 78.08 % nitrogen, 20.95 % oxygen, 0.93 % argon, 0.0018 % neon, 0.00052 % helium, 0.00011 % krypton, and 0.0000087 % xenon, with small amounts of CO₂, H₂, CH₄, NO, and O₃.
In nuclear power plants, krypton can also be harvested from uranium fission. Unlike atmospheric krypton, fission yields both stable and radioactive isotopes.
The Manufacturing Process
Making Liquid Air
- Air is first filtered to remove particulates and then treated with an alkali to absorb water and CO₂.
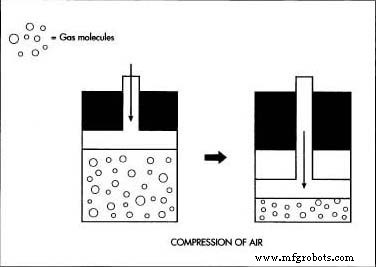
- The dry air is compressed at high pressure, raising its temperature; it is then cooled by refrigeration.
- During the subsequent expansion into a chamber, the sudden pressure drop absorbs heat, cooling the air to about –321 °F (–196 °C) and liquefying most atmospheric gases.
Separating the Gases
- Gases with very low boiling points—helium, hydrogen, and neon—are removed directly.
- Fractional distillation exploits the distinct boiling points of remaining gases.
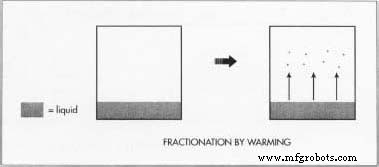
- As the liquid air warms, argon, oxygen, and nitrogen vaporize first; krypton and xenon stay liquid until higher temperatures.
Separating Krypton from Xenon
- Liquid krypton and xenon are adsorbed onto silica gel or activated charcoal and then subjected to further fractional distillation.
- Warming the mixture releases krypton as a gas while xenon remains liquid due to its higher boiling point.
- Krypton is purified by passing it over hot titanium, which removes all non‑noble elements.
Separating Krypton Isotopes
- For applications requiring a single isotope, thermal diffusion separates krypton by density differences.
- A heated wire in a vertical glass tube establishes convection, carrying lighter isotopes to the top for collection.
Packaging and Shipping
- Krypton is sealed in Pyrex bulbs at atmospheric pressure or in steel canisters under high pressure. Its inertness makes it safe, non‑toxic, non‑explosive, and non‑flammable, eliminating special shipping requirements.
Quality Control
Purity is verified through spectroscopic analysis: the gas is heated, and the emitted light is dispersed by a prism or grating to produce a spectrum. Sharp, bright lines characteristic of krypton confirm purity; any deviation signals contamination.
Byproducts and Waste
Fractional distillation yields large quantities of nitrogen (≈75 % of air), which is essential for producing ammonia and protecting materials from oxidation. Liquid nitrogen is used in cryopreservation and refrigeration.
Oxygen, making up about 20 % of air, is consumed heavily by the steel industry for oxidation of carbon and by environmental applications such as sewage treatment and waste incineration. Liquid oxygen also serves as rocket propellant.
Other noble gases—argon, neon, and xenon—find niche uses: argon in light bulbs and as a shielding gas; neon in iconic signage; xenon in strobe lights and high‑intensity lamps.
The Future
The future supply of krypton may hinge on the trajectory of nuclear power. As a fission byproduct, nuclear reactors could become a significant source. However, if fusion or alternative energy sources supplant fission, atmospheric extraction will likely remain the dominant method.
Manufacturing process
- What is VMC Machining? An Expert Overview of Vertical Machining Centers
- Expert Guide to Aluminum Laser Marking: Precision, Durability, and Industry Applications
- MIG vs. TIG Welding: Selecting the Right Arc Welding Technique for Your Project
- Comprehensive Guide to Laser Marking: Types, Benefits, and Applications
- Key Considerations for High‑Volume Swiss Machining
- CNC Prototyping Guide: Precision, Speed, and Cost‑Effective Production
- Expert Shaft Manufacturing: Precision Processes & Custom Solutions
- Fiber Laser Marking: Precision, Speed, and Versatility for Modern Industries
- Electropolishing vs. Passivation: Choosing the Right Stainless Steel Finishing
- Krypton: Properties, Production, and Uses



