Nano Gold Clusters: A Powerful Catalyst for Oxidation Reactions
 Nanosized gold clusters
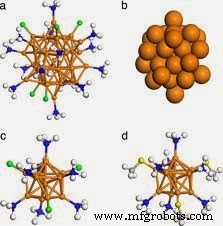
Nanosized gold clusters are celebrated for their ability to catalyze a wide range of reactions—including oxidations, esterifications, and epoxidations. Yet, the origins of the metal’s remarkable reactivity remained elusive. Notably, gold’s role as a catalyst for CO oxidation has long been recognized. Recent computational work now reveals that CO itself can act as a co‑catalyst, boosting the oxidation of CO to CO₂ when adjacent to gold nanoclusters.
Mechanism
Researchers from the University of Nebraska and Xiangtan University in China have elucidated a self‑oxidation pathway. Their findings show that when CO binds to specific triangular Au₃ active sites on gold nanoclusters in the presence of O₂, the CO molecule assists in cleaving an adjacent OCOO intermediate. The study demonstrates that an Au₃‑bound CO neighbor accelerates O–O bond scission, yielding two CO₂ molecules.
Nanosized gold clusters
Nanosized gold clusters are celebrated for their ability to catalyze a wide range of reactions—including oxidations, esterifications, and epoxidations. Yet, the origins of the metal’s remarkable reactivity remained elusive. Notably, gold’s role as a catalyst for CO oxidation has long been recognized. Recent computational work now reveals that CO itself can act as a co‑catalyst, boosting the oxidation of CO to CO₂ when adjacent to gold nanoclusters.
Mechanism
Researchers from the University of Nebraska and Xiangtan University in China have elucidated a self‑oxidation pathway. Their findings show that when CO binds to specific triangular Au₃ active sites on gold nanoclusters in the presence of O₂, the CO molecule assists in cleaving an adjacent OCOO intermediate. The study demonstrates that an Au₃‑bound CO neighbor accelerates O–O bond scission, yielding two CO₂ molecules.
Nanomaterials
- Nano Tungsten Carbide – A Platinum‑Like Catalyst
- Gold: History, Properties, Mining, and Future Applications
- Plasmonic Nanoparticles: Harnessing Surface Plasmons for Advanced Photothermal Applications
- Nano Hydroxyapatite: Bioactive Material for Advanced Biomedical Applications
- Build an Arduino Nano-Based Eating Robot – Step-by-Step Guide
- Ultrasonic Tripwire Sensor System with Arduino Nano
- Gold Anodizing Explained: Durable Gold Finish for Base Metals
- AuNi18: High-Performance Gold Vacuum Brazing Alloy
- 30% Cold-Worked Gold (Au): Enhanced Strength, Ductility & Alloying Potential
- Gold (Au) 50% Cold-Worked: Enhanced Strength & Versatile Alloying



