How Metal Powders Are Made: A Comprehensive Guide to Preparation Methods

How Metal Powders Are Made: A Comprehensive Guide to Preparation Methods
Metal powder is the cornerstone of powder metallurgy, enabling the creation of complex components through molding and sintering. It is indispensable across machinery, aerospace, chemical processing, and advanced composites. Understanding how these powders are produced—whether by mechanical disintegration, atomization, or chemical synthesis—is essential for engineers, material scientists, and manufacturers seeking high‑performance parts.

Illustration of metal powder production methods
A typical metal powder consists of particles smaller than 1 mm, with size ranges classified as follows:
- 1000–50 µm: Conventional powder
- 50–10 µm: Fine powder
- 10–0.5 µm: Very fine powder
- <0.5 µm: Ultrafine powder
- 0.1–100 nm: Nanopowder
Primary Production Routes
Industrial metal powders are mainly produced via two overarching categories: mechanical methods and physicochemical methods. These encompass direct refinement from solid, liquid, or gaseous states and reduction, pyrolysis, or electrochemical pathways for metal compounds.
Refractory metal compounds such as carbides, nitrides, borides, and silicides are typically produced through compounding or reduction‑combination processes. The resulting powder’s morphology and particle size vary significantly with the method employed.

Comparison of metal powder production techniques
1. Mechanical Production
Mechanical routes fracture metal into fine particles using external forces while preserving chemical composition. The most prevalent techniques are ball milling and gas‑assisted grinding, both offering simplicity and high throughput.
Ball Milling
Ball milling operates through rolling or vibratory action, applying varying strain rates to break and refine metal particles. It is well suited for alloys such as Sb, Cr, Mn, and Fe‑Cr, enabling continuous operation, high yield, and applicability to both dry and wet grinding.
Key advantage: scalable production of ultrafine powders from high‑melting‑point metals. Limitation: material selectivity is low, and real‑time particle classification is challenging.

TEM images of ball‑milled powder samples after 12h, 18h, and 24h at 150 rpm
Gas‑Assisted Grinding
In this method, compressed gas is sprayed through a nozzle into a grinding chamber, inducing high‑energy collisions that pulverize the material. The resulting particles are then classified by a turbine classifier; coarse fragments are recirculated until the desired size is achieved.
Benefits include dry processing—eliminating dehydration steps—yielding high‑purity, finely distributed powders with smooth surfaces. Drawbacks: high gas consumption (often inert gas or nitrogen) and elevated manufacturing costs, making it best suited for brittle metals and alloys.
Atomization
Atomization fragments molten metal or alloy using high‑pressure gas, liquid, or a high‑speed blade to produce fine droplets that solidify in a collector. The process introduces no chemical change, preserving the metal’s intrinsic properties.
It is the primary method for producing powders of Fe, Sn, Zn, Pb, Cu, and alloy systems such as bronze, brass, and steel. Atomized powders exhibit high sphericity, controllable particle size, low oxygen content, and competitive production costs.
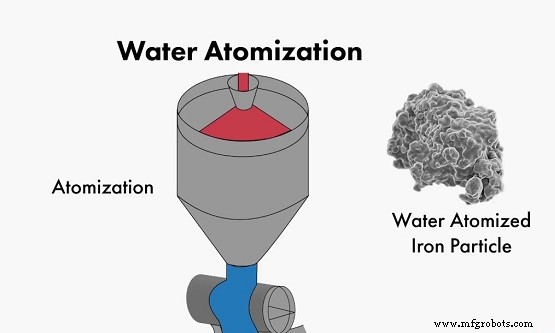
Atomization process illustration
Challenges include lower throughput for ultrafine powders and higher energy consumption compared to mechanical methods.

Microstructure of 3D‑printed stainless steel powder
2. Physicochemical Production
Physicochemical routes manipulate the chemical state or aggregation of raw materials to yield ultrafine powders. The principal approaches are reduction, electrolysis, hydroxyl (metal‑carbonyl) decomposition, and chemical replacement.
Reduction
This technique reduces metal oxides or salts using hydrogen, solid carbon, or metal‑based reducing agents. Hydrogenation is especially common: the metal forms a hydride, which is mechanically pulverized, and residual hydrogen is removed under vacuum.
Typical applications: Ti, Fe, W, Mo, Nb, and W‑Re alloys. Example: titanium reacts violently with hydrogen above 2.3 % H₂, producing a loose hydride that can be ground into fine powder and then dehydrogenated at ~700 °C.
Pros: straightforward operation, high efficiency, low cost. Cons: limited to metals that readily form hydrides and become brittle during absorption.
Electrolysis
Electrolysis deposits metal onto a cathode from an aqueous or molten salt solution, yielding powders with purity up to 99.7 % or higher. It also allows precise particle‑size control, enabling ultrafine powders.
Commonly used for Cu, Ni, Fe, Ag, Sn (aqueous) and Zr, Ta, Ti, Nb (molten salts). Main drawback: significant energy consumption and higher operational costs.
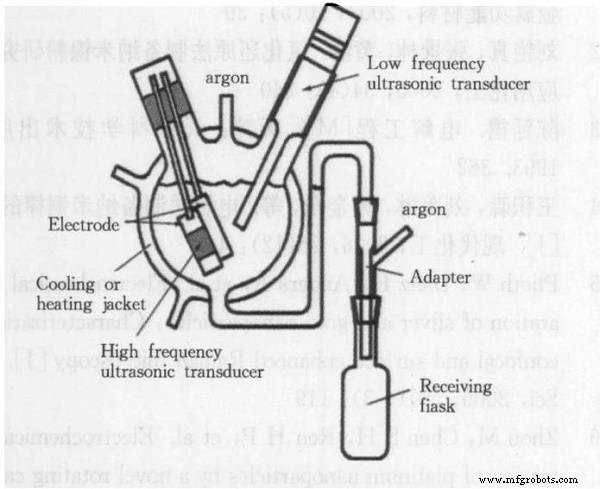
Ultrasonic electrolysis device for iron powder
Hydroxyl (Metal‑Carbonyl) Decomposition
This method synthesizes metal carbonyls (e.g., Ni(CO)₄, Fe(CO)₅) from metal salts and CO, then thermally decomposes them to produce fine, high‑purity powders. It is particularly effective for nickel and iron, and for alloy powders such as Fe‑Ni, Fe‑Co, and Ni‑Co.
The powders are exceptionally fine but the process is capital intensive.
Chemical Replacement
Also known as displacement, this approach substitutes a less active metal from a salt solution with a more reactive metal. The resulting metal is then further refined. It is suited for inert metals like Cu, Ag, and Au.
Key Takeaways
Metal powder production spans a spectrum of methods, each tailored to specific metals, desired particle sizes, and purity requirements. Mechanical routes excel in high‑yield, cost‑effective production of high‑melting‑point alloys, while physicochemical processes offer superior purity and fine‑particle control for specialized applications.
For further insights into refractory metal powders—such as tungsten and tantalum—visit Advanced Refractory Metals (ARM), a leading global supplier headquartered in Lake Forest, California.
Metal
- Spherical Tungsten Powder Production Techniques: Methods, Advantages, and Industry Applications
- Professional Guide to Producing High‑Purity Rhenium Powder
- How Tantalum Powder is Produced: Grades, Properties, and Manufacturing Techniques
- TZM Alloy: Properties, Applications & Advanced Manufacturing Techniques
- Molybdenum Powder: Types, Properties, and Industrial Applications
- Tungsten Powder: Applications, Production Methods, and Industry Insights
- Rhenium Metal Powder: Applications in Superalloys, Catalysts, and Precision Instruments
- A Comprehensive Guide to Metal 3D Printing Technologies
- 11 Proven Metal Casting Techniques for Precision Manufacturing
- Smithy Secrets: Mastering Modern Forging Techniques



